 |
A 28-year-old male presented to the office complaining of painless vision loss in his left eye for the past week. He can still see, but notices distortions in his vision. At first, he attributed his problem to extreme amounts of reading and other near work leading to “eye strain.” He thought that things would get better on their own, but when he noticed the doorframe in his home appeared ballooned out “like a pincushion,” he immediately sought care.
Examination
His entering uncorrected visual acuities were 20/25 OD and 20/40 OS, with both eyes improving to 20/20 with pinhole. Subsequent refraction also yielded 20/20 acuity in each eye, but with a -0.50 DS lens for the right eye and a +0.1.00 DS for the left.
The remainder of his external examination was normal and a dilated fundus examination showed all his right eye’s structures were normal; however, a large, bullous sensory macular detachment was visible in the left eye. He was subsequently diagnosed with central serous chorioretinopathy (CSC).
With a diagnosis in hand, he was further questioned regarding possible etiologies.
He denied use of any type of steroid but, when asked about his vocation, he explained that he was a law school graduate under tremendous anxiety as he studied for the bar exam.
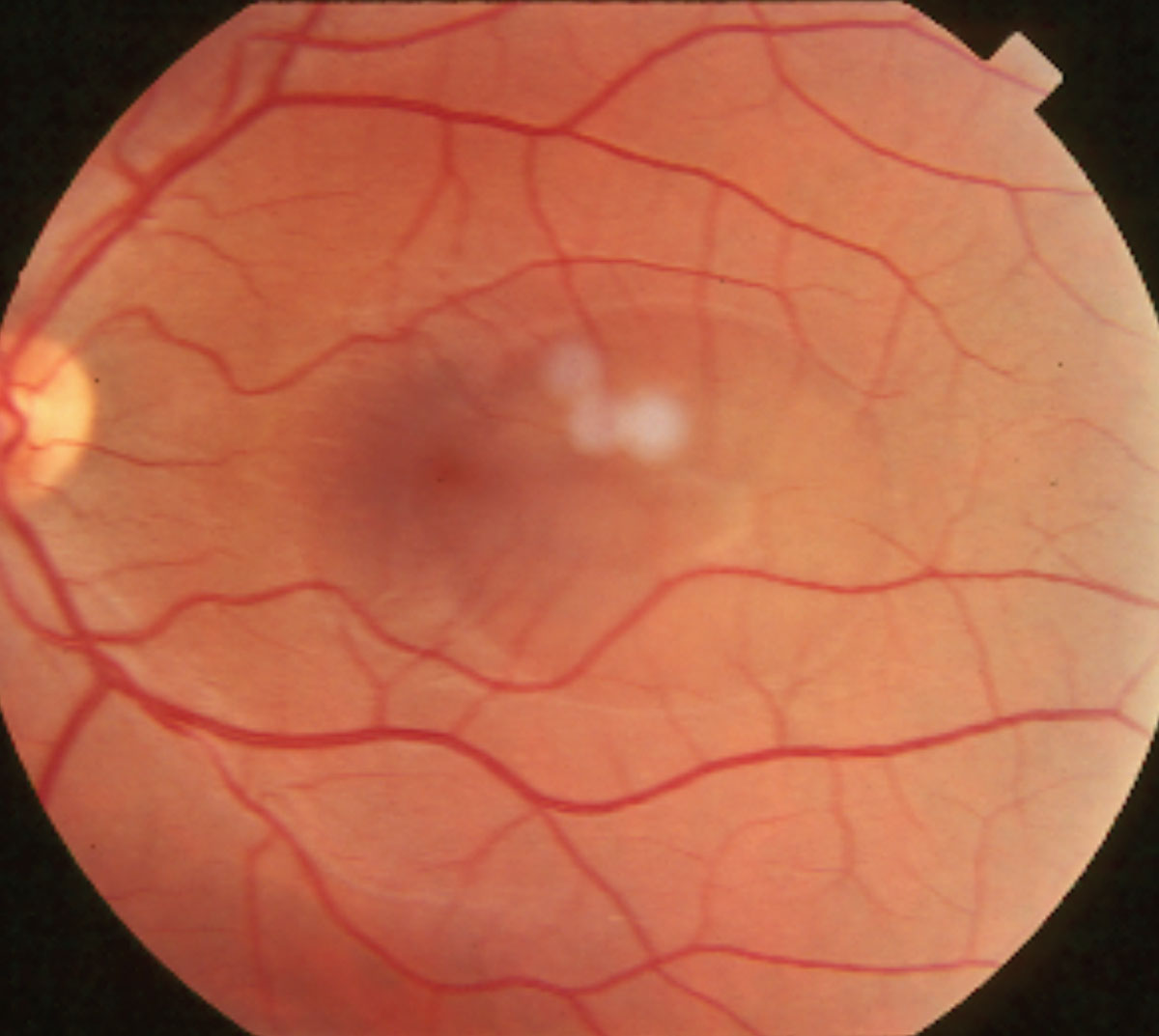 |
| This patient’s fundus photo shows a large paracentral central serous chorioretinopathy. This pathology can cause sudden-onset blurred vision and decreased color perception in addition to other complaints. |
Discussion
Patients with CSC usually present with complaints of sudden onset distortion or blurring of central vision. They may report metamorphopsia, decreased color perception or even a relative central scotoma.1-3 Patients often report a history of using corticosteroids (topical, injectable or oral), sympathomimetic agents or medications for erectile dysfunction.1-9 If such a history is not offered, it is crucial to specifically ask about these associations. Other contributory elements may include antibiotics, uncontrolled hypertension, alcohol, allergic respiratory disease and obstructive sleep apnea.2,10 However, steroids are the greatest exogenous precipitating factor.2
Patients with CSC are typically between the ages of 25 and 50.11,12 Men are afflicted far more frequently than women, with an incidence ratio of about 6:1.1,2,12-15 Perhaps the most well-known association with CSC is the psychological profile known as “type A” personality. These individuals, who are described as exhibiting the characteristics of time urgency, aggressiveness, hostility and competitiveness, seem to be particularly predisposed to developing CSC.16-19
Mild hyperopic refractive shift (+1.25D or less) is often noted in the affected eye. Funduscopic exam shows a distinct, round or oval serous elevation of the macula with a loss of the foveal light reflex. An underlying area of retinal pigment epithelium (RPE) detachment may be seen concurrently in about 10% of patients, and this may easily be missed without optical coherence tomography (OCT).20,21 The possibility of choroidal neovascularization (CNV) exists as well, and these cases are typically associated with a poor visual outcome.22,23
CSC appears to have a multifactorial etiology, with various systemic associations and a complex pathogenesis. The primary dysfunction appears to be localized ischemia or inflammation at the level of the choriocapillaris, which leads to hyperpermeability; this in turn results in decompensation of the RPE, causing a focal detachment of the overlying neurosensory retina.19,20 Biochemical changes are likely at the root of this process. In patients with CSC, serum levels of catecholamines and glucocorticoids appear elevated, directly influencing the integrity of Bruch’s membrane.17-19,24 Stimulation of adrenergic receptors often results in release of secondary messengers (e.g., cyclic adenosine monophosphate), and this may produce the vascular or RPE changes that result in CSC.25
Therapies
Most cases of CSC are fortunately self-limiting over a period of three to 12 months.1,11 The prognosis for visual recovery is excellent, with most regaining their pre-event acuity. Upon diagnosing the condition, any corticosteroid therapy should be immediately discontinued, if possible, as 90% of CSC cases resolve spontaneously following the cessation of steroids.26 While the acute phase of CSC is usually self-limiting, the condition may be recurrent in as many as 50% of affected individuals.27
In non-remitting or recurrent cases, focal laser photocoagulation has been use in an attempt to arrest the leakage.28 However, focal laser therapy does not necessarily ensure improvement in visual acuity; it merely hastens recovery and possibly diminishes the likelihood of recurrence.1,28 There are risks associated with this treatment, most notably laser damage to the fovea and subsequent CNV formation.1,23,24 For these reasons, most will employ laser therapy only in cases that fail to respond within a reasonable period of time, recurrent cases or cases in which the patients are overtly symptomatic and insist on definitive treatment. Others will avoid laser for fear of further disrupting the RPE/Bruch’s complex and have felt that this treatment is like “putting fertilizer on a weed.”
Photodynamic therapy (PDT) with Visudyne (verteporfin, Bausch + Lomb) has also been used successfully in the treatment of CSC; research shows it improves visual acuity, reduces leakage on fluorescein angiography, reduces subretinal fluid as demonstrated by OCT, and fosters choroidal remodeling with decreased choroidal permeability.29,30 While PDT has promising results, this treatment has limitations. First, CSC is not an approved use for PDT; thus, most insurances will not cover a treatment costing approximately $2,000. Also, the laser used is no longer sold or readily available. No compelling evidence at this time shows intravitreal injections of anti-VEGF drugs improves outcomes in CSC.31
Future Therapy
A potential therapy in the pipeline is the potassium-sparing diuretic Aldactone (spironolactone, Pfizer), a mineralocorticoid receptor antagonist. Aldactone is used to treat primary hyperaldosteronism, in which the body produces excess amounts of the hormone aldosterone, regulating sodium and water levels. Aldactone treats fluid retention in people with congestive heart failure, cirrhosis of the liver and nephrotic syndrome. This medication is also used to treat or prevent hypokalemia.
In a study of acute CSC patients treated with spironolactone (40mg orally, twice daily) for two months compared with an observational group, investigators saw a faster resolution in the orally treated group.32 Additionally, in eyes with persistent CSC, Aldactone therapy was associated with a statistically significant improvement in subretinal fluid as well as visual acuity.33 Another report confirmed a positive effect of spironolactone in non-resolving CSC and good results in patients with recurrent CSC who responded to spironolactone initially.34
It appears that a good clinical response and low cost makes Aldactone a reasonable option for patients with CSC. There are, however, some adverse effects including decreased libido and male gynecomastia. In that CSC often has a good prognosis for spontaneous recovery, these effects must be considered.
The patient presented here, once educated on his treatment options, elected to be observed only. The good news was that over several weeks, the CSC lesion resolved without complications commensurate with his passing the bar. The bad news was that he plans to specialize in malpractice litigation. Sigh.
1. Ross A, Ross AH, Mohamed Q. Review and update of central serous chorioretinopathy. Curr Opin Ophthalmol. 2011;22(3):166-73. 2. Haimovici R, Koh S, Gagnon DR, et al. Risk factors for central serous chorioretinopathy: a case-control study. Ophthalmology. 2004;111(2):244-9. 3. Kleinberger AJ, Patel C, Lieberman RM, Malkin BD. Bilateral central serous chorioretinopathy caused by intranasal corticosteroids: a case report and review of the literature. Laryngoscope. 2011;121(9):2034-7. 4. Baumal CR, Martidis A, Truong SN. Central serous chorioretinopathy associated with periocular corticosteroid injection treatment for HLA-B27-associated iritis. Arch Ophthalmol. 2004;122(6):926-8. 5. Fernandez CF, Mendoza AJ, Arevalo JF. Central serous chorioretinopathy associated with topical dermal corticosteroids. Retina. 2004;24(3):471-4. 6. Koyama M, Mizota A, Igarashi Y, et al. Seventeen cases of central serous chorioretinopathy associated with systemic corticosteroid therapy. Ophthalmologica. 2004;218(2):107-10. 7. Fraunfelder FW, Fraunfelder FT. Central serous chorioretinopathy associated with sildenafil. Retina. 2008;28(4):606-9. 8. Gordon-Bennett P, Rimmer T. Central serous chorioretinopathy following oral tadalafil. Eye (Lond). 2012;26(1):168-9. 9. Michael JC, Pak J, Pulido J, et al. Central serous chorioretinopathy associated with administration of sympathomimetic agents. Am J Ophthalmol. 2003;136(1):182-5. 10. Kloos P, Laube I, Thoelen A. Obstructive sleep apnea in patients with central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2008;246(9):1225-8. 11. Marcuson J, Riley T. Central serous chorioretinopathy. Optometry. 2008;79(5):241-51. 12. Kitzmann AS, Pulido JS, Diehl NN, et al. The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology. 2008;115(1):169-73. 13. Todd KC, Hainsworth DP, Lee LR, et al. Longitudinal analysis of central serous chorioretinopathy and sex. Can J Ophthalmol. 2002;37(7):405-8. 14. How AC, Koh AH. Angiographic characteristics of acute central serous chorioretinopathy in an Asian population. Ann Acad Med Singapore. 2006;35(2):77-9. 15. Desai UR, Alhalel AA, Campen TJ, et al. Central serous chorioretinopathy in African Americans. J Natl Med Assoc. 2003;95(7):553-9. 16. Yannuzzi LA. Type A behavior and central serous chorioretinopathy. Trans Am Ophthalmol Soc. 1986;84:799-845. 17. Wynn PA. Idiopathic central serous chorioretinopathy--a physical complication of stress? Occup Med (Lond). 2001;51(2):139-40. 18. Zakir SM, Shukla M, Simi ZU, et al. Serum cortisol and testosterone levels in idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 2009;57(6):419-22. 19. Gemenetzi M, De Salvo G, Lotery AJ. Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye (Lond). 2010;24(12):1743-56. 20. Mudvari SS, Goff MJ, Fu AD, et al. The natural history of pigment epithelial detachment associated with central serous chorioretinopathy. Retina. 2007;27(9):1168-73. 21. Chang MA, Bressler SB. Photodynamic therapy for chronic pigment epithelial detachment in central serous chorioretinopathy. Can J Ophthalmol. 2009;44(2):221-2. 22. Shanmugam MP, Bhende M. Retinal pigment epithelial tears associated with idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 2000;48(4):315-7. 23. Konstantinidis L, Mantel I, Zografos L, Ambresin A. Intravitreal ranibizumab in the treatment of choroidal neovascularization associated with idiopathic central serous chorioretinopathy. Eur J Ophthalmol. 2010;20(5):955-8. 24. Chan WM, Lai TY, Liu DT, Lam DS. Intravitreal bevacizumab (avastin) for choroidal neovascularization secondary to central serous chorioretinopathy, secondary to punctate inner choroidopathy, or of idiopathic origin. Am J Ophthalmol. 2007;143(6):977-983. 25. Sun J, Tan J, Wang Z, et al. Effect of catecholamine on central serous chorioretinopathy. J Huazhong Univ Sci Technolog Med Sci. 2003;23(3):313-6. 26. Sharma T, Shah N, Rao M, et al. Visual outcome after discontinuation of corticosteroids in atypical severe central serous chorioretinopathy. Ophthalmology. 2004;111(9):1708-14. 27. Wong R, Chopdar A, Brown M. Five to 15 year follow-up of resolved idiopathic central serous chorioretinopathy. Eye (Lond). 2004;18(3):262-8. 28. Lim JW, Kang SW, Kim YT,et al. Comparative study of patients with central serous chorioretinopathy undergoing focal laser photocoagulation or photodynamic therapy. Br J Ophthalmol. 2011;95(4):514-7. 29. Taban M, Boyer DS, Thomas EL, et al. Chronic central serous chorioretinopathy: photodynamic therapy. Am J Ophthalmol. 2004;137(6):1073-80. 30. Chan WM, Lam DS, Lai TY, et al. Choroidal vascular remodelling in central serous chorioretinopathy after indocyanine green guided photodynamic therapy with verteporfin: a novel treatment at the primary disease level. Br J Ophthalmol. 2003;87(12):1453-8. 31. Salehi M, Wenick AS, Law HA, et al. Interventions for central serous chorioretinopathy: a network meta-analysis. Cochrane Database Syst Rev. 2015 Dec 22;(12):CD011841. 32. Sun X, Shuai Y, Fang W, et al. Spironolactone versus observation in the treatment of acute central serous chorioretinopathy. Br J Ophthalmol. 2018;102(8):1060-5. 33. Falavarjani KG, Amirsardari A, Habibi A, et al. Visual and Anatomical Outcomes of Spironolactone Therapy in Patients with Chronic Central Serous Chorioretinopathy. J Ophthalmic Vis Res. 2017;12(3):281-9. 34. Herold TR, Rist K, Priglinger SG, et al. Long-term results and recurrence rates after spironolactone treatment in non-resolving centralserous chorio-retinopathy (CSCR). Graefes Arch Clin Exp Ophthalmol. 2017;255(2):221-9. |

