When managing ocular disease with systemic medications, clinicians must select drugs and dosages that are both safe and effective—and the process becomes much more complicated when the patient is a child. We must give special consideration to this vulnerable patient group, as certain medications considered safe in the adult population may be contraindicated in children. Additionally, the pathogens most common in adult infectious disease may be different in this population, requiring the use of an antibiotic different from what is used in the adult population for similar-appearing infectious ocular disorders.1
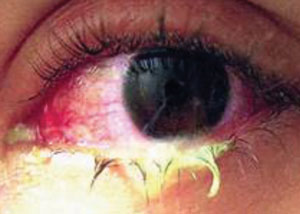 | |
| H. Influenzae, a gram-negative bacteria, is a common cause of conjunctivitis. |
Dosing Dilemmas
Choosing the correct dose of a systemic medication for the pediatric population is not a simple linear calculation based on the size of an individual; instead, one must consider differences in pharmacokinetics—the ability to absorb, distribute, metabolize and excrete a medication—to individualize the treatment to the patient. For example, a 13-year-old may be tall for his age, yet still have the liver maturity of a young teenager. Similar to other special populations such as the elderly, pregnant patients and those with diabetes, the clinician must first choose a drug that is safe for children and then carefully calculate a modified dosage. This modification is dependent on several factors, including age, weight, body surface area, liver and kidney function and pre-existing systemic disease.1,2
There are three popular dosing calculations. Clark’s rule was an attempt to adjust dosage strictly on weight and was used for individuals weighing less than 100lbs (40kg). Because it neither takes into account factors such as liver and kidney function nor is specific enough for many of the potent drugs used today, its use should be avoided. Its greatest flaw is seen in overweight children for whom the calculated dosage would be well above the appropriate amount.
Fried’s rule, used in infants, is based on age only and does not take into account the variability in children’s sizes or the pharmacokinetics of the prescribed medication.
The third formula uses body surface area (BSA) in square meters to calculate IV dosages in infants. The calculation is a ratio of the child’s BSA divided by an ideal adult’s BSA. That ratio is multiplied by the average adult dose to produce a reduced dose for the child. It is a complicated calculation that is prone to error.3
The most precise calculation is based upon the recommendations developed by the drug companies themselves. These recommendations are listed in the package insert and are available via many drug databases. These calculations are always in metric measurements and usually in mg/kg of body weight. Most importantly, these dosages also list a maximum pediatric dose. Note that the recommended dose may be listed for each administration of drug, given a certain number of times per day, or as a 24-hour dose that you must divide by the recommended frequency of daily dosing. Pay strict attention to the exact language to avoid dosing errors.
Indications
Except for degenerative conditions, such as AMD, children present with many of the same conditions as adults. Ocular disorders include acute infectious disease, allergic (atopic) disease, acute injuries and inflammation. By far, the most common conditions in children involve treatment with antibiotics. Most acute ocular disease is treated with topical agents. The following conditions commonly need systemic therapy with or without topical agents.
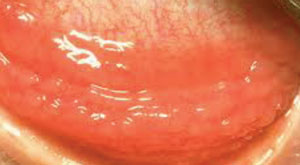 | |
| Chlamydia is a common cause of ophthalmia neonatorum. |
• Conjunctivitis. Although not normally treated with systemic medications, the most common cause of acute infectious conjunctivitis in children is the gram-negative pathogen Haemophilus influenzae. This is different from adults, who most commonly present with adenoviral conjunctivitis. Haemophilus colonizes children in their upper respiratory tract, from which it travels via the eustachian tube to the middle ear (otitis media). It can travel via the nasolacrimal system to the eye to cause conjunctivitis, to the lung to produce pneumonia and to the CNS to cause meningitis.
The introduction of a Haemophilus vaccine has reduced the incidence from more than 40% of children’s eye disease to approximately 32%. It still remains the major cause of acute infectious eye disease. Because there is a reservoir of bacteria, children colonized by Haemophilus who show evidence of systemic infection (e.g., otitis media, fever) should be treated both topically and systemically.9
• Ophthalmia neonatorum. Any ocular infection within the first 30 days of life is known as ophthalmia neonatorum. The patient must be tested to rule out Neisseria infections. Currently, the most common cause of ophthalmia neonatorum is chlamydia, which must be managed with systemic therapy. If chlamydia is present secondary to maternal transfer, the mother and her partner should be treated as well.
Closely related to chlamydia is trachoma, which causes inflammation and scarring of the conjunctiva and cornea. Trachoma represents a major cause of vision loss worldwide, though far less so in the United States and other developed countries. The infection is spread via flies, with children as the primary reservoir.
Another condition, preseptal cellulitis, is commonly caused by injury or insect bites, and must be differentiated from orbital cellulitis.9 Orbital cellulitis, a deeper, more serious condition, is almost always caused by an adjacent severe sinusitis. The hallmarks of orbital cellulitis are decreased ocular motility and globe displacement, often proptosis. This can be life-threatening because of the orbit’s proximity to the brain, and should be considered a true emergency requiring immediate IV antibiotics and hospitalization.9
Common pathogens that can produce both preseptal and orbital cellulitis include Staphylococcus species, both aureus and epidermidis, as well as Streptococcal species. Strep. is particularly dangerous in that it produces the enzymes streptokinase and hyaluronidase, allowing it to spread through tissues in a disease process known as erysipelas.9
• Bacterial dacryocystitis/dacryoadenitis. Characterized by acute or chronic infection or inflammation of the lachrimal sac, dacryocystitis is not uncommon in newborns with lacrimal stenosis prior to repair or spontaneous resolution of the blockage. Complete or partial obstruction of the duct can lead to tear stasis and opportunistic infection. The acute version is most commonly seen in children. The route of infection can be blood-borne, transconjunctival, transneuronal or via direct trauma. Common bacteria that cause dacryoadenitis include Staph. sp., Strep. sp., Haemophilus influenzae and Neisseria.
| Drug Testing in Children Before 1970, children and pregnant women were never involved in drug studies, leading to a lack of knowledge of how children process important medications. By the mid ’70s, the American Academy of Pediatrics (AAP) argued for the right to include children in well-controlled studies, stating that physicians were forced, without proper data, to conduct uncontrolled experiments on kids when they prescribed medications that had never been tested in this population. In 1977, guidelines established by the AAP stated that children could participate in minimal risk studies if one parent gave permission or higher risk studies with the permission of both parents.4-8 |
Treatment with broad-spectrum antibiotics should be initiated until cultures, titers and lab studies identify the specific etiology. Appropriate treatment choices include:
- Amoxicillin/clavulanate: 25mg to 40mg/kg/D in a BID divided dose
- Cefprozil: 15mg/kg/D Q12H, with a maximum dose of 1gm/D
- Azithromycin: 10mg/kg/D x 3D, maximum daily dose of 400mg
- Clarithromycin: 7.5mg/kg/D BID9
• Ocular rosacea. More commonly affecting the central face (i.e., nose and cheeks) than the eyes and adnexa, rosacea is an inflammatory dermatologic condition that has a variety of trigger mechanisms, including spicy foods, cold weather, sun exposure and stress. Rosacea is classified into four subtypes by the National Rosacea Society: erythematotelangectatic, papulopustular, phymatous and ocular. It is common for patients to express more than one type, and more than 50% of patients with rosacea also present with ocular rosacea.9
Ocular rosacea causes chronic inflammatory changes of the eyelid margins, leading to an uneven “wiper effect” of the eyelid against the corneal surface, as well as poor meibomian function with tear film instability and evaporative dry eye. The inflammatory mediators released from the lid also commonly lead to conjunctivitis and keratitis.9
The cornerstone of medical therapy for ocular rosacea has been the tetracyclines and the macrolide antibiotics, which have been shown clinically effective in reducing signs and symptoms of both facial and ocular rosacea. However, tetracyclines are absolutely contraindicated in younger children. Their use produces permanent tooth discoloration and bone abnormalities. In children older than age 12, the dose of doxycycline is 50mg to 100mg BID. Fortunately, children younger than 12 can use the macrolides. The newest broad-spectrum macrolide agents include clarithromycin (Biaxin, Abbott Laboratories) and azithromycin (Zithromax, Pfizer). Clarithromycin is usually dosed in individuals that weigh less than 40kg at 7.5mg/kg BID. Because azithromycin has a 68-hour half-life, it is dosed at 10mg/kg/D for only three days per week. Treatment commonly lasts for months.9
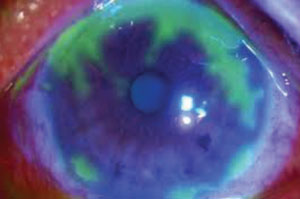 | |
| Ocular manifestations of HSV-1 include: blepharitis, conjunctivitis, corneal epithelial disease and stromal disease. |
• Herpes simplex. The majority of the population will be exposed to herpes simplex virus (HSV) by a young age. Transmission of the virus occurs by direct contact with infected lesions, salivary droplets, fomites and healthy, asymptomatic carriers. The incubation period between contact and expression is approximately three to nine days.
While the primary infection may be mild or subclinical in nature, the virus may reactivate and ultimately involve various ocular structures. Potential manifestations include blepharitis, conjunctivitis, corneal epithelial disease, stromal disease, neurotrophic ulceration, endotheliitis, trabeculitis, uveitis, retinitis, or a combination of these. Active viral replication, pathogenicity of the virus and host immune responses are major factors influencing disease recurrence.9
There are two types of HSV that affect humans. HSV-1 commonly causes labialis ocular manifestations, while HSV-2 is usually genital in nature and is acquired by sexual contact or, in the case of children, is acquired during vaginal delivery. The specific treatment is dependent on the tissues affected. Ocular epithelial disease is not treated with steroids, whereas inflammatory forms include topical and systemic antiviral therapy and topical steroids.9
When prescribing oral antivirals, the dosage depends on the agent. The usual adult dose of acyclovir is 400mg five times daily. Pediatric dosing using the 200mg/5cc suspension is 10mg/kg Q8H.
• Varicella zoster virus (VZV). Varicella is the primary infection from VZV, resulting in inflammatory as well as immunologic reactions. VZV is responsible for the systemic varicella infection (chickenpox) upon initial exposure to the virus and for herpes zoster (shingles) upon reactivation. The primary VZV infection results in an exanthematous reaction of the skin resulting in the formation of pustules (“pocks”) that contain live virus. The lesions are typically self-contained and tend to resolve over a period of one to two weeks. The incidence of childhood VZV has decreased due to the vaccine.
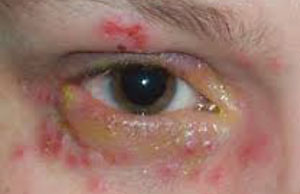 | |
| Chickenpox often involves ocular hyperemia with a watery discharge. |
Typically, ocular involvement results in mild to moderate ocular hyperemia with a watery discharge. Symptoms include foreign body sensation, occasionally blurred vision and photophobia. Signs can include a dendriform keratitis and are distinctly different from those seen in herpes simplex. Treatment is supportive, although more severe cases may require oral acycylovir. Pediatric dosing using the 200mg/5cc suspension is 10mg/kg Q8H.9
• Allergy/immune disease. Typical seasonal allergic eye disease is managed with topical agents. However, children experiencing significant ocular anaphylactic symptoms (i.e., moderate to severe immediate hypersensitivity reactions triggered by exposure to antigens such as animal dander, peanuts or medications that commonly include penicillins, sulfonamides and opiates) may require systemic intervention. Children with asthma are particularly vulnerable to significant allergic events. The speed of onset of antihistamine agents is directly related to their lipid solubility. Unfortunately, highly lipid-soluble agents also cross the blood-brain barrier and can lead to significant anticholinergic side effects. These include sedation as well as dry eye and mouth symptoms. The most important rapid-onset oral antihistamine is diphenhydramine. The less sedating agents, unfortunately, have a much slower onset of action and are reserved for prophylactic use.
Diphenhydramine, the drug of choice for acute mast cell mediated allergic reactions, is available in over-the-counter adult and pediatric forms. The pediatric syrup is available with 12.5mg/5cc. The pediatric dosage is 1mg/kg/D up to TID, with a maximum of 50mg/D. Overdosage can lead to convulsions and seizures.
Pharmacology
Knowing more about the agents you are prescribing to your pediatric patients can help ensure the most appropriate dosage. Here are many of the common systemic medications used in the pediatric population.
• Penicillins. Amoxicillin is a semisynthetic antibiotic with a broad spectrum of bactericidal activity against many gram-positive and gram-negative microorganisms. It is, however, susceptible to degradation by β-lactamases, and the spectrum of activity does not include organisms that produce these enzymes. Clavulanic acid is a β-lactam structurally related to the penicillins that can inactivate a wide range of β-lactamase enzymes commonly found in microorganisms resistant to penicillins and cephalosporins. In particular, it has good activity against the clinically important plasmid-mediated β-lactamases frequently responsible for transferred drug resistance.10
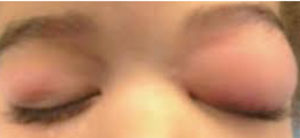 | |
| Ocular anaphylaxis is often treated with a rapid-onset oral antihistamine. |
The amoxicillin and clavulanic acid formulation in Augmentin (GlaxoSmithKline) protects amoxicillin from degradation by β-lactamase enzymes and effectively extends the antibiotic spectrum of amoxicillin to include many bacteria normally resistant to amoxicillin and other β-lactam antibiotics. Thus, Augmentin possesses the properties of a broad-spectrum antibiotic and a β-lactamase inhibitor. Amoxicillin/clavulanic acid has been shown to be active against most strains of bacteria that produce pediatric ocular disease, including Staph. and Strep. species and, most importantly, Haemophilus influenzae.11
• Macrolides. Azithromycin represents the new broad-spectrum keto-macrolide antibiotics. They act by binding to the 50S ribosomal subunit of susceptible microorganisms and interfering with microbial protein synthesis. Nucleic acid synthesis is not affected. It is bactericidal and effective against Staph. sp., Strep. sp. and, most importantly in children, Haemophilus. This structural change increases their spectrum of activity over erythromycin. The keto-macrolide agents also include clarithromycin.
Azithromycin differs from clarithromycin in that it has a much longer half-life; therefore, the dosing schedule is markedly different from antibiotics that are used for seven to 10 days. Most pediatric dosing regimens of azithromycin last for only three days, with the drug remaining active in the body for over 10 days. This long half-life improves compliance and negates the need for prescription refills. The most important parameter for safe use is proper pediatric dosing following the guidelines based on age and weight. Overdosage affects the liver, and children are particularly susceptible because of their relative reduced renal function, so patients with liver disease should be prescribed a markedly reduced dosage. The macrolides are particularly valuable as a replacement for individuals allergic to penicillins.
• Cephalasporins. Cefprozil is an oral second-generation cephalosporin. The second-generation drugs are particularly valuable in the pediatric population for two reasons: (1) They are less susceptible to destruction from penicillinase-producing bacteria, and (2) they show greater activity against Haemophilus influenzae. The recommended oral dose of cefprozil is 15mg/kg/dose BID. It is available in liquid dosage forms of 125mg/5ml and 250mg/5ml. The maximum dose is 1gm/day.
| Recommended Pediatric Dosages11 Diphenhydramine: 2-5yrs: 6.25mg every 4-6 hours, max 24 hour dose of 37.5mg/24 hours 6-11yrs: 12.5 to 25mg every 4-6 hours, max 24 hour dose of 150mg/24 hours >11yrs: 25-50mg every 4-6 hours, maximum of 300mg/24 hours Prednisone/prednisolone: 1mg/kg/day divided up to QID/24 hours Augmentin: 25mg/kg/day in divided dose of two to three times/day Cefprozil: 15mg/kg/dose BID Azithromycin: 10mg/kg daily for three days Clarithromycin: 7.5mg/kg/dose BID Doxycycline: 2.1mg/kg/dose BID only in children 8 years or older Acyclovir: 10 to 20mg/kg/dose every eight hours Acetaminophen: 15-20mg/kg/dose PRN every 4 hours. Maximum of 5 doses; not to exceed75mg/kg/day or 2.6g total/day Ibuprofen: 10mg/kg every 6 hours, max dose of 800mg not recommended for children under 6 months Acetaminophen with codeine: 0.5-1mg/kg of codeine component every 4 hours PRN; caution in children under 3 years of age Acetazolamide: 5mg/kg/dose every 6-12 hours/24 hours |
• Tetracyclines. Recently, the use of doxycycline in the management of ocular disease has focused on its ability to reduce ocular inflammation. The indications for its use include ocular rosacea, dry eye, recurrent erosion and bacterial keratitis. A recent study by ASCRS recommends it be used in place of topical steroids for reduction of inflammation secondary to certain forms of bacterial keratitis.12 The AAP recommends its use for ages eight and older. The major adverse effect of this class of drug in younger children is permanent tooth staining and abnormal bone development. The recommended oral dose for children ages eight and older is 2.1mg/kg/D BID. It should never exceed the adult dose, which is 100mg BID.
• Antiviral therapy. Acyclovir is a synthetic purine nucleoside analog with in vitro and in vivo inhibitory activity against HSV-1, HSV-2 and VZV. The inhibitory activity of acyclovir is highly selective due to its affinity for the enzyme thymidine kinase (TK) encoded by HSV and VZV. This viral enzyme converts acyclovir into acyclovir monophosphate, a nucleotide analog. The monophosphate is further converted into diphosphate by cellular guanylate kinase and into triphosphate by a number of cellular enzymes.
In vitro, acyclovir triphosphate stops replication of herpes viral DNA by: (1) competitive inhibition of viral DNA polymerase, (2) incorporation into and termination of the growing viral DNA chain, and (3) inactivation of the viral DNA polymerase. The greater antiviral activity of acyclovir against HSV compared to VZV is due to its more efficient phosphorylation by the viral TK.
The major potential adverse effect is crystalline nephropathy. It is important to calculate a proper dosage and further ensure patients have normal renal function and remain well hydrated during therapy. The drug is extremely safe when the prescriber and parents are conscious of these factors. Of the three major oral antiviral medications—acyclovir, valacyclovir and famcyclovir—only acyclovir is available in a pediatric syrup of 200mg/5cc.
• Prednisone/prednisolone. The steroid prednisone is popular by virtue of its ability to rapidly control both acute and chronic inflammatory disease. In children it is commonly used to manage acute, type 1 allergic events and asthma. It is used chronically for the management of rheumatoid disorders. Ocular inflammatory conditions, including uveitis and allergic eye and periocular skin reactions (hives), also respond well to short-term steroid use. The most important contraindication to even short-term use in any population is diabetes, as glucocorticoids can produce a dramatic rise in blood glucose levels. The dosage for all age groups is 1mg/kg/day in divided dosage three to four times daily with a maximum dosage of 60mg/D. It is available in tablets that range from one to 100mg/dose and a pediatric syrup that contains 5mg/5ml (Pediapred).
Pain Management
It is always important to consider the management of pain associated with ocular injury or inflammation. Adjunctive pain management is most important in those children who may not be able to clearly express their level of discomfort. Dacryocystitis, preseptal cellulitis, corneal abrasion and ocular burns are only a few indications that require short-term pain management. Safety is a major consideration in children managed with analgesics. Liver and kidney damage as well as neurologic adverse effects can occur with inappropriate use of these agents. These side effects must be considered when prescribing these agents. A few pain management options include:
• Acetaminophen. This OTC drug has long been considered the analgesic of choice for management of pain in special populations such as children and pregnant patients.
For the last several years, the FDA has mounted a significant public campaign to educate both the public and the pharmaceutical industry on the dangers of acetaminophen overdose.
Acetaminophen acute toxicity is a major cause of acute hepatic failure in children. The reasons for overdose include children assuming fruit-flavored syrups and chewable tablets are “candy,” and the lack of understanding that using multiple OTC products containing acetaminophen contributes to a cumulative daily dose that exceeds the current recommended maximum adult dose of 2.6g/D, leading to a toxic cumulative dose.
Advise parents to note the presence of acetaminophen in all products given to their children. The current recommended dose is 15mg/kg every four to six hours PRN for pain with a maximum of five doses/24 hours, not to exceed 75mg/kg/day or 2.6g total/day.
• Ibuprofen. An oral NSAID, ibuprofen is available in a pediatric dosage of 100mg/5ml. The recommended dose is 10mg/kg/dose every six hours. Do not exceed an average adult dose of 600mg QID. Also, ibuprofen is not recommended in children under the age of six months.
The major contraindication to the use of any NSAID such as ibuprofen is bleeding, as these drugs inhibit platelet activity and increase the risk of hemorrhage in those with injuries, abnormal clotting or in the presence of other drugs that have anti-coagulant properties.
• Opiates. The most common form of opiate analgesic drugs is a combination agent. Of the common combination agents, acetaminophen with codeine is, by far, the most popular. The use of acetaminophen with codeine is synergistic for pain management; that is, both drugs enhance the other’s ability to reduce pain without enhancing toxicity. Each drug in a combination dosage form must be judged separately for its safety in each patient. In addition to acetaminophen’s effect on the liver, there is a significant risk of allergic reaction to codeine. All opiates demonstrate cross-sensitivity, so a patient allergic to one opiate is allergic to all of them. Opiates also can produce significant respiratory depression, which is particularly dangerous in children with a history of asthma. The drug can produce anticholinergic side effects, including marked sedation.
The recommended dose of acetaminophen with codeine is based on the codeine content of the medication and should not exceed 0.5mg to 1mg/kg/dose every four hours PRN pain. The liquid dosage form contains 12mg of codeine and 120mg of acetaminophen per 5ml. The dosage is 0.2ml to 0.4ml/kg/dose every four hours PRN pain. The product should be used with extreme caution in children under the age of three.
When treating ocular disease in the pediatric population, always weigh the need for systemic therapy against the associated risks. Specifically, one must determine, based on the clinical history, the specific drug, dosage and frequency of use that will be safe and effective for that particular patient.
Dr. Onofrey is a clinical professor and the executive director of continuing education programs at the University of Houston.
1. Bartelink IH, Rademaker MA, Schobben A, et al. Guidelines on pediatric dosing on the basis of developmental physiology and pharmacokinetic considerations. Clinical Pharmacokinetics. 2006 Nov;45(11):1077-97.2. Harris M, Patterson J, Morse J. Doctors, nurses, and parents are equally poor at estimating pediatric weights. Pediatr Emerg Care. 1999;15(1):17-8.
3. Moore P. Children are not small adults. Lancet. 1998;352(9128):630.
4. United States Government Accountability Report to Congressional Committees. Pediatric Drug Research Studies Conducted Under Best Pharmaceutical for Children Act. March 2007.
5. Rodriguez W, Selen A, Avant D, et al. Improving pediatric dosing through pediatric initiatives: what we have learned. Pediatrics. 2008;121(3):530-9.
6. American Academy of Pediatrics Committee on Drugs. Guidelines for the ethical conduct of studies to evaluate drugs in pediatric populations. Pediatrics. 1995;95(2):286-94.
7. Christensen ML, Helms RA, Chesney RW. Is pediatric labeling really necessary? Pediatrics. 1999;104(3):593-7.
8. Specific requirements on content and format of labeling for human prescription drugs: revision of “pediatric use” subsection in the labeling. 59 Federal Register. 1994;64240.
9. Onofrey BE, Skorin L, Holdeman NR. The Ocular Therapeutics Handbook: A Clinical Manual. 3rd ed. Philadelphia: Lippincott; 2012.
10. Onofrey BE. Clinical Optometric Pharmacology and Therapeutics. Philadelphia: Lippincott; 1996.
11. www.drugs.com.
12. Solomon R, Donnenfeld E, Holland E, et al. Microbial keratitis trends following refractive surgery: Results of the ASCRS infectious keratitis survey and comparisons with prior ASCRS surveys of infectious keratitis following keratorefractive procedures. J. Cataract and refractive surgery. 2011;37:1343-50.

