Normal tension glaucoma (NTG) is among the greatest enigmas in eye care. In fact, some believe that as a separate diagnosis, it is a distinction without a difference. Those who prefer to characterize NTG as a separate entity point to evidence of alterations of blood flow to the eye among NTG patients, for example. This may help to explain why patients with intraocular pressure (IOP) in the normal range suffer glaucomatous damage.
As we learn more about the pathogenesis of glaucoma in general, some new ideas emerge. Maybe the statistically normal range of pressure is an erroneous construct. Perhaps the answer is perfusion pressure to the optic nerve head (ONH). Maybe the mechanism is related to an imbalance of intracranial pressure (ICP), which is indexed to cerebrospinal fluid pressure (CSFP). Could it be that the IOP climbs above the normal range at other times than when sampled in the office setting? Could NTG be related to peripheral vascular dysregulation, anatomical variations or even genetics? Is normal tension glaucoma a unique diagnosis or an overlap? All of these processes have been proposed over the years.
The distinction between the subset of open-angle glaucoma patients who have consistently “normal” intraocular pressure (IOP) and those whose IOP is measured above that upper limit is completely arbitrary. That population may represent between 20% and nearly 40% of cases of diagnosed glaucoma in the United States.1 The notion of pressure-independent damage consistent with a diagnosis of glaucoma suffered at IOP within the statistically normal range has been a difficult diagnosis for many years.2 Early and ongoing studies on IOP-lowering strategies for stalling progression have offered management guidance.
Potential Contributors to NTG
• Low optic nerve perfusion pressure (either situational or continuous)
• Low blood pressure
• Low cerebrospinal fluid pressure
• Situationally elevated IOP
• Systemic vascular dysregulation signs (e.g., cold extremities, migraine sufferer)
The Collaborative Normal Tension Study Group (CNTGSG) evaluated whether IOP should be reduced in patients with NTG. After the five-year trial, CNTGSG results found that 12% (7/61) of medically or surgically treated patients vs. 35% (31/79 controls) of untreated patients exhibited visual field or optic disc progression.3 Their results showed a positive benefit from a 30% reduction of IOP with disease progression. Even though the treated group benefited from the lower IOP, there was still progression among those patients.
Should we lower IOP further than 30%? This study was performed before prostaglandins and prostamides were approved for treatment of glaucoma. Therefore, further IOP reduction was difficult to obtain at the time of the study.
Later, CNTGSG published a study looking at the medical history of the untreated patient to investigate a common factor among the NTG patients. History of hypertension, undergoing major surgery, migraine and non-migraine headaches, and family history of stroke, glaucoma and diabetes were all reported as common factors. However, females with a history of migraines were identified to have a great risk of developing rapid visual field deterioration.4 Perhaps we should be looking more carefully at vascular regulation of blood flow to the optic nerve.
A more recent work—the Low-Pressure Glaucoma Study (LoTGS)—looked at visual field progression with monotherapy of brimonidine tartrate 0.2% vs. timolol maleate 0.5% (both dosed BID).5 Despite similar average IOP-lowering effects from both drugs, the brimonidine treatment group had a much lower rate (9.1%) of visual field progression vs. the timolol group (39.2%).
The mystery remained: If IOP is lowered by the same amount, why the difference in progression? A review of the evidence has shown that alpha-2 adrenergic agonists may confer neuroprotective properties in experimental optic nerve injuries.6 Evidence for such efficacy in human glaucoma damage is lacking.

Reproduced from Kamal D, Hitchings R. Normal tension glaucoma – a practical approach. Br J Ophthalmol 1998;82:835–840 with permission from BMJ Publishing Group Ltd.
Before enumerating potential connections with NTG, let’s look at the differential diagnoses. A comprehensive approach includes complete ocular and systemic histories. For example, has there ever been a history of blunt trauma or an episode of acute blood loss?
Our clinic serves a patient who, as a teenager, severed his right radial artery. This required hospitalization, but his optic nerve on that side is markedly more damaged than the fellow eye. Despite consistently very low IOP, he has shown slow progression over two decades of follow-up.
Systemically, episodes of acute (e.g, accident or surgery requiring blood transfusion) or chronic blood loss (e.g, bleeding ulcer) may be important contributors to observed disc damage that may be stable or result in a vulnerable ONH. In these situations with statistically normal IOP, appropriate ancillary testing may prove a better clinical course than potentially invasive testing. Ischemic optic neuropathies (e.g., NAION, GCA) are other masqueraders of NTG with distinct clinical characteristics that would require a separate description beyond this discussion.
Definitive guidance for neuroimaging considerations in questionable cases of NTG has been reported.7 Those of younger age at initial observation (<50 years), with decreased visual acuity (VA poorer than 20/40), specific visual-field defects respecting the vertical midline or pallor greater than cupping are the ideal candidates for such investigations.
Blood Flow Regulation and Dysregulation
Much of the evidence favoring a distinct category for NTG comes from blood flow studies. These include the notion of peripheral vascular dysregulation, as evidenced by cold extremities, specific measures of nail fold capillary blood flow, the cold pressor test and careful blood flow measurements of the retinal and choroidal blood flow.8-10
The cellular and molecular considerations in these scenarios are beyond the scope of this article and involve oxidative mechanisms and nitric oxide regulation, for example. The interested reader is referred to a review on the topic published earlier this year.10
Perhaps related to glaucoma in general is the idea of the balance between IOP opposing perfusion pressure to the optic nerve head. Mean perfusion pressure has been linked to progressive glaucomatous damage in the Barbados eye study, an association reviewed recently.11 Mean perfusion pressure links the parameters of systolic and diastolic blood pressure with measured IOP.
One surrogate that may be applied to clinical situations is the diastolic ocular perfusion pressure (DOPP). Simply subtracting the IOP from the diastolic BP gives this result. The range of normal or threshold for safety remains elusive, however. In a practical sense, the lower the value, the more likely the risk for hypoperfusion.

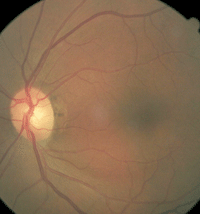
Optic nerve head images of a 72-year-old black female who has been followed at UAB Eye Care for over 30 years. Since her previous visit a year earlier, she developed an inferior rim notch in the right eye. There is corroborating evidence from imaging and visual field data. She was initiated on IOP-lowering treatment. The fellow eye did not show change. Note the disc asymmetry.
The evidence presented here suggests that low perfusion pressure is associated with greater and more progressive glaucomatous damage, making the case for hypoperfusion as the most likely candidate, but perhaps not the only one, to explain observed glaucomatous damage in the absence of elevated IOP.11 So, as clinicians we should pay attention to the IOP and BP among our NTG patients and those suspected of glaucoma in general.
Other recent evidence implicates low nocturnal ocular perfusion pressure as a risk factor for NTG.12 This study incorporated 24-hour IOP and blood pressure monitoring to provide a profile of the interaction. The results support the evidence favoring a defective autoregulatory mechanism among NTG patients. Interestingly, this report is from a non-ophthalmic publication. The idea of relative ischemia as a contributor to optic nerve deterioration, however, is not new in the ophthalmic literature. The idea had been suggested more than two decades earlier.13
Going forward, if we can merge the data from these two measurements, perhaps a clearer picture of glaucomatous damage at “normal” intraocular pressure will emerge.
Most recently a distinction between ocular and systemic blood flow parameters of normal subjects and those with either early NTG or POAG has been reported.14 Interestingly, the authors reported that among a broad panel of ocular and systemic findings there is considerable overlap between both glaucoma groups in the early stages of these diseases but distinct from normal subjects serving as controls. While the vascular alterations described are not those routinely measured, they included peripheral arterial stiffness, carotid intima-media thickness and ocular perfusion pressure.
The authors conclude that NTG and POAG are points on a continuum rather than distinct entities.
CSF pressure
It is well known that increased intracranial pressure results in the clinical observation of a swollen optic disc, or papilledema. What could be the implication of reduced intracranial pressure? The notion of a connection to glaucoma has been crystalized recently.15 In summary, it looks something like this: The combined influences of IOP, systemic blood pressure and cerebrospinal fluid pressure (CSFP) interact at the level of the lamina cribrosa. Measuring only the transcorneal pressure (IOP) as is done clinically may be inadequate to explain glaucomatous damage and NTG in particular.
Mentioned earlier was the influence of reduced ocular perfusion, an emerging measure and influence in glaucoma (both high- and low-pressure). The model includes the presence of low systemic blood pressure, either in general or situationally at night, being associated with low CSFP, which produces an abnormally high trans-laminar pressure diffential. This scenario is similar to the condition of CSFP in the normal range with the IOP being elevated as a component of the mechanism of glaucomatous damage. This model is proposed to explain why patients with NTG have low systemic blood pressure, and why eyes with normal- and high-pressure glaucoma may develop similar optic disc damage patterns.
Management of NTG
Whether one believes that NTG is a separate entity, emerging evidence points to potential new management strategies beyond lowering IOP. Traditional (in-office, snapshot) measurement of IOP is inadequate to characterize its day-long, 24-hour behavior. Similarly, a single blood pressure measurement does not characterize a patient’s blood pressure during the night or when performing other activities. Introducing CSFP into the mix may help to explain damage at (or to) the level of the lamina cribrosa.
Any discussion
of NTG must include optic disc hemorrhages. The observation of an optic
disc hemorrhage has been associated with glaucoma, and NTG specifically,
to the extent that these are referred to eponymously as Drance
Hemorrhages in tribute to Stephen Drance who established this
connection.20 The flame- or splinter-shaped hemorrhage crosses the disc
margin and has been associated by some with a greater likelihood of
progression. Other studies, however, show little difference in visual
field deterioration, but greater structural change (i.e., RNFL loss).21
Recently,
a report of sophisticated measures between NTG and HTG patients has
suggested that NTG patients may show more central visual field damage.22
The authors suggest considering a 10-2 visual field testing protocol to
measure progression. Stereometric parameters measured with the
Heidelberg Retinal Tomograph-III, however, did not distinguish between
the HTG and NTG groups.22 Another recent report has suggested that
systemic hypertension in a setting of optic disc hemorrhage is
associated with NTG.23 The confusion surrounding risk of progression
(vs. a sign consistent with a diagnosis) has a number of explanations,
including the particular population studied, the stage of disease at
initial intervention, means of measurement, and so on.
The interaction among all three of these parameters may give clinicians an improved picture of progression in glaucomatous damage (especially NTG). How will this be done? Documenting a 24-hour blood pressure profile is possible; these data may be available from the patient’s cardiologist, for example. Technology for 24-hour IOP measurement is currently in clinical trials in the US, although no device is currently FDA approved. Look to the future for clinical applications. Patterns in this paradigm may not be consistent, leading to confusion or explanation of variability.16 Finally, incorporating the influence of CSFP, while perhaps the most tedious to measure clinically, may allow more information on glaucomatous damage and especially that in NTG.
The Optic Nerve in NTG
Concretely, it has been known for many years that the topical beta-blockers are relatively ineffective when administered at night. Systemic absorption may reduce systemic blood pressure and, by extension, perfusion to the optic nerve. Therefore, especially in susceptible individuals, topical beta-blockers should not be administered at night.17,18 In fact, in those glaucoma patients treated for systemic hypertension with documentation of progression in the face of “controlled” IOP, the prescriber should be consulted with the evidence.11-13
The future is bright for our ability to better characterize glaucomatous damage, regardless of whether the IOP is outside or within the statistically normal range. Given a better knowledge of the factors surrounding and producing glaucomatous damage will also give us more effective tools for preventing or minimizing it. Apart from the traditional management of glaucoma (lowering IOP), however, no consistent evidence for any alternative strategy to preserve structure and function in NTG has emerged victorious.
Connecting the Dots Clinically
The normal IOP range is a statistically defined as ranging from roughly 10mm to 21mm Hg. We know that some patients will tolerate a higher IOP without damage while others will show progressive damage when the IOP is within the normal range. Clearly, pressure-independent factors are involved. As mentioned above, these may include situational elevations of IOP, systemic vascular regulation factors, the influence of systemic medications and most recently reported, CSFP. Given our current inability to continuously measure IOP, looking at other clinical measures such as 24-hour blood-pressure monitoring and CSFP may be useful but tedious in the normal clinical setting.
One suggestion has been to observe spontaneous venous pulse at the optic nerve head. The results of a recent study using color Doppler imaging to measure retinal vessel blood velocities showed that, on average, glaucoma patients had lower retinal vein velocities.19 Combined with the lower prevalence of spontaneous venous pulsation among glaucoma patients, especially NTG, the implication is that this may represent altered hemodynamics within the eye and orbit. If one accepts this premise, then the influence of reduced CSFP rises in significance as a probable metric for glaucomatous damage and perhaps an index of the likelihood of progression.
Dr. Semes is a professor of optometry at the University of Alabama School of Optometry. He also serves as Director, Externship Programs. Dr. Reid is an assistant professor of optometry at UAB.
1. Sommer A, Tielsch JM, Katz J, et al. Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans. The Baltimore Eye Survey. Arch Ophthalmol. 1991;109:1090-5.
2. Kamal D, Hitchings R. Normal tension glaucoma – a practical approach. Br J Ophthalmol. 1998;82:835–40.
3. Collaborative Normal-Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Am J Ophthalmol. 1998;126: 498-505.
4. Anderson DR; Normal Tension Glaucoma Study. Collaborative normal tension glaucoma study. Curr Opin Ophthalmol. 2003;14:86-90.
5. Krupin T, Liebmann JM, Greenfield DS, Rosenberg LF, Ritch R, Yang JW; Low-Pressure Glaucoma Study Group. The Low-pressure Glaucoma Treatment Study (LoGTS) study design and baseline characteristics of enrolled patients. Ophthalmology. 2005 Mar;112: 376-85.
6. Saylor M, McLoon LK, Harrison AR, Lee MS. Experimental and clinical evidence for brimonidine as an optic nerve and retinal neuroprotective agent: an evidence-based review. Arch Ophthalmol. 2009 Apr;127(4):402-6. doi: 10.1001/archophthalmol.2009.9.
7. Greenfield DS, Siatkowski RM, Glaser JS, et al. The cupped disc: Who needs neuroimaging? Ophthalmology. 1998;105:1866-74.
8. Flammer J, Haefliger IO, Orgül S, Resink T. Vascular dysregulation: a principal risk factor for glaucomatous damage? J Glaucoma. 1999; 8:212-9.
9. Flammer J, Orgül S, Costa VP, et al. The impact of ocular blood flow in glaucoma. Prog Retin Eye Res. 2002; 21:359-93.
10. Mozaffarieh M, Flammer J. New insights in the pathogenesis and treatment of normal tension glaucoma. Curr Opin Pharmacol. 2013;13:43-9.
11. Leske MC. Ocular perfusion pressure and glaucoma: clinical trial and epidemiologic findings. Curr Opin Ophthalmol. 2009;20:73-8.
12. Ramli N, Nurull BS, Hairi NN, Mimiwati Z. Low nocturnal ocular perfusion pressure as a risk factor for normal tension glaucoma. Prev Med. 2013 Jan 23. pii: S0091-7435(13)00020-0. doi:10.1016/j.ypmed.2013.01.007. [Epub ahead of print]
13. Hayreh SS, Zimmerman MB, Podhajsky P, Alward WL. Nocturnal arterial hypotension and its role in optic nerve head and ocular ischemic disorders. Am J Ophthalmol. 1994;117:603-24.
14. Mroczkowska S, Benavente-Perez A, Negi A, Sung V, Patel SR, Gherghel D. Primary open-angle glaucoma vs normal-tension glaucoma: the vascular perspective. JAMA Ophthalmol. 2013;131:36-43.
15. Jonas JB, Wang N. Intracranial pressure and glaucoma. J Glaucoma. 2013;22 Suppl 5:S13-4.
16. Mansouri K, Liu JH, Weinreb RN, Tafreshi A, Medeiros FA. Analysis of continuous 24-hour intraocular pressure patterns in glaucoma. Invest Ophthalmol Vis Sci. 2012; 53:8050-6.
17. Hayreh SS. Effect of nocturnal blood pressure reduction on retrobulbar hemodynamics in glaucoma. Graefes Arch Clin Exp Ophthalmol. 2002;240:867-8.
18. Hayreh SS, Zimmerman MB, Podhajsky P, Alward WL. Nocturnal arterial hypotension and its role in optic nerve head and ocular ischemic disorders. Am J Ophthalmol. 1994;117:603-24.
19. Abegão Pinto L, Vandewalle E, De Clerck E, Marques-Neves C, Stalmans I. Lack of spontaneous venous pulsation: possible risk indicator in normal tension glaucoma? Acta Ophthalmol. 2012 Jul 9. doi: 10.1111/j.1755-3768.2012.02472.x.
20. Drance SM. Some factors in the production of low tension glaucoma. Br J Ophthalmol. 1972; 56: 229-42.
21. Kim SH, Park KH. The relationship between recurrent optic disc hemorrhage and glaucoma progression. Ophthalmology. 2006;113: 598-602.
22. Thonginnetra O, Greenstein VC, Chu D, Liebmann JM, Ritch R, Hood DC. Normal versus high tension glaucoma: a comparison of functional and structural defects. J Glaucoma. 2010;19:151-7.
23. Kim YD, Han SB, Park KH, et al. Risk factors associated with optic disc haemorrhage in patients with normal tension glaucoma. Eye (Lond). 2010;24:567-72.
24. Anderson DR, Drance SM, Schulzer M; Collaborative Normal-Tension Glaucoma Study Group. Natural history of normal-tension glaucoma. Ophthalmology. 2001;108: 247-53.
25. Drance SM. What can we learn from the disc appearance about the risk factors in glaucoma? Can J Ophthalmol. 2008 Jun;43(3):322-7.

