 |
A 36-year-old white male presents to your office with a chief complaint of intermittent pain on the right side of his head and orbital area along with redness and foreign body sensation in his right eye. As a construction worker, he’s concerned that he may have gotten something in his eye.
External examination shows a mild ptosis (1.5mm) with minimal palpebral injection and no lid edema in the right eye. No foreign body of the cornea or bulbar conjunctiva is noted on a slit lamp examination, nor is any evident on lid eversion. The cornea is clear, the anterior chamber is well formed and quiet. His unaided visual acuity is 20/20 each eye.
Pupil testing reveals anisocoria greatest under dim illumination and most noticeable during the first few seconds after the lights were turned down. The right pupil showed a delay in dilation consistent with “dilation lag” found in Horner’s syndrome. So, you perform bilateral tactile assessment of the forehead to discern potentially uneven sweating patterns. Anhidrosis is not evident and not reported by the patient.
But what if your office setting has no available pharmacological agents able to confirm or localize Horner’s syndrome? Should the patient be referred for diagnostic testing, or should imaging be ordered? If so, what is the best approach to ordering imaging for acute onset Horner’s syndrome?
This article offers several pearls on the differential diagnosis and timely management of acute-onset Horner’s syndrome patients.
The Classic Triad
Patients who present with unilateral ptosis, miosis and anhidrosis have the classic triad of signs associated with oculosympathetic paresis, also known as Horner’s syndrome.1 Because the oculosympathetic pathway of the eye is rather long and at times winding, the list of potential causes that can disrupt the efferent signal along this pathway is long.
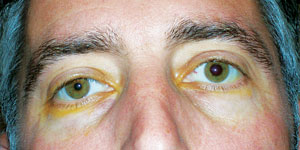 | |
| Anisocoria in a patient with recent onset of Horner’s syndrome. Photo: Alan G. Kabat, OD. |
When disruption along the oculosympathetic pathway occurs, the efferent signal to Mueller’s muscle and the iris dilator muscle is adversely affected, resulting in pseudo-enophthalmos (1mm to 2mm ptosis of the upper lid coupled with reverse ptosis of the lower lid) and a miotic pupil, respectively. The ptosis and anisocoria can be quite subtle and may go unnoticed by friends, family and coworkers.
The pupil is miotic because the circumferential sphincter muscle of the iris, which is innervated by the parasympathetic pathway, is no longer equally opposed by the oculosympathetic pathway, which innervates the radial dilator muscle.2 The resultant anisocoria is most noticeable under dim illumination. Dilation lag is a classic sign of Horner’s syndrome—when the slit lamp beam is turned off, the anisocoria will be most noticeable during the first four or five seconds of viewing. The abnormal pupil will slowly dilate or “lag behind” over 10 to 15 seconds, making the pupil asymmetry less evident. This partially explains why some cases of Horner’s syndrome are diagnosed during a routine eye examination.
Causes
Horner’s syndrome is most often due to a benign etiology; but a few sinister causes require urgent diagnosis and care. These include, but are not limited to:
- Spontaneous or traumatic carotid artery dissection.
- Neoplasia in the neck.
- Lung apex (Pancoast tumor).
- Childhood neuroblastoma.
Establishing the underlying diagnosis of Horner’s syndrome begins with a detailed clinical history, timely pharmaceutical testing and diagnostic imaging.
Is It Long Standing?
Long-standing cases are less concerning than acute cases. Inspecting old photographs may confirm longstanding Horner’s syndrome and support the decision to monitor. The presence of iris heterochromia (affected iris is blue and the other brown) certainly indicates that the lesion occurred somewhere between birth and two years of age. The most common cause is birth trauma resulting in a brachial plexus injury.
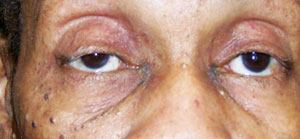 | |
| New onset right ptosis (with miosis) in a patient who presented with Horner’s syndrome secondary to a Pancoast tumor. Photo: Alan G. Kabat, OD. |
Is It Iatrogenic?
Patients who have had head, neck or chest surgery may be experiencing a temporary or permanent side effect from the procedure. The preganglionic neuron is the most common site of injury for iatrogenic Horner’s syndrome. Such procedures include coronary artery bypass surgery, carotid endarterectomy or stenting, insertion of a pacemaker, epidural anesthesia, chest tube insertion, as well as lung or mediastinal surgery. If the patient reports recent surgery involving one or more these anatomical areas, be sure to notify the surgeon of your findings.
Is There Ipsilateral Anhidrosis?
Anhidrosis, or loss of hemifacial sweating, may be seen in a subset of Horner’s patients. The level of involvement ranges from barely detectable to striking. The area of involvement may include the brow, forehead or the entire side of the head and face, depending on lesion location. If a patient has a lesion near the common carotid artery, loss of sweating involves the entire side of the face. With lesions distal to the carotid bifurcation, the lack of sweating is typically confined to the medial aspect of the forehead and side of the nose.
A patient may report an exertion-induced asymmetric reaction of the skin remaining pale and dry on the side of the face with the affected pupil. The presence of anhidrosis narrows the list of potential causes to the central and preganglionic pathways.
Pain Matters
If Horner’s syndrome is associated with acute-onset ipsilateral head, orbital, face or neck pain, consider it an internal carotid artery dissection (ICAD) until proven otherwise.3 Studies show that a partial-Horner’s syndrome is found in 36% to 58% of all ICAD cases.4 ICAD is rare, with an incidence of three in 100,000 and is classified as either spontaneous (i.e., connective tissue disease) or traumatic.5 While the most common traumatic cause of ICAD is motor vehicle accidents, any exertional force that induces a twisting of the head or neck may cause the lamina intima of the ICA to tear secondarily to hyperextension, rotation, or both.3,6 This can lead to bleeding into the laminar wall of the vessel, resulting in ischemia or thrombosis. If not treated with anticoagulation therapy in a timely fashion, the patient may suffer stroke or even death.6,7
| Testing Hierarchy for Horner’s Syndrome | |||
| First-Order Neuron Lesions Occurs uncommonly in isolation, usually one of a number of neurologic findings. | |||
| Associated Findings • Dysphagia (difficulty swallowing) • Dysarthria (slowed or slurred speech) • Hemisensory loss • Ataxia (lack of muscle coordination) • Vertigo • Nystagmus | Diagnostic Tests • Cocaine test (+) • Iopidine test (+) • Paredrine (+) | Anatomic Sites • Hypothalamus, thalamus, brainstem • Cervical spinal cord | Imaging • CT of the chest & CT / CTA of the head & neck or • MRI of the chest & MRI / MRA of head & neck |
| Second-Order Neuron Lesions Most often caused by trauma or tumor, including malignant tumors. | |||
| Associated Findings • Hx of prior head, face, or neck trauma • Previous thoracic or neck surgery / chest tube or catheter placement • Any facial, neck, axillary, shoulder or arm pain • Chronic cough or hemoptysis (coughing up blood or blood stained mucous) | Diagnostic Tests • Cocaine test (+) • Iopidine test (+) • Paredrine (+) | Anatomic Sites • Cervicothoracic spinal cord • Brachial plexus • Anterior aspect of the Neck • Lung apex • Mediastinum | Imaging • CT of the chest & CT / CTA of the head & neck or • MRI of the chest & MRI / MRA of head & neck |
| Third-Order Neuron Lesions Variable causes from benign to life-threatening. | |||
| Associated Findings • Diplopia (i.e. VI N palsy) • V1 & V2 numbness • Pain | Diagnostic Tests • Cocaine test (+) • Iopidine test (+) • Paredrine (-) | Anatomic Sites • Superior cervical ganglion • Internal carotid artery • Cavernous sinus • Orbital apex | Imaging • MRI / MRA of the head & neck |
If, on the other hand, Horner’s syndrome is associated with a radiating pain from the shoulder to the ulnar side of the arm and hand, this suggests an apical lung tumor 55% to 60% of the time.8 Pancoast tumors are also associated with neck pain as well as parasthesias of the hand.9 More than 200,000 new cases of lung cancer are diagnosed every year, approximately 5% of which are Pancoast tumors.10 Incidence is highest in males between the age of 40 and 60 with a history of smoking.10 Pancoast tumors are a form of non-small-cell carcinoma, most commonly with squamous cell carcinoma (45% to 50%).8
No Cocaine? No Worries!
Pharmacologic testing may be helpful in diagnosing and localizing a Horner’s syndrome lesion.
Four percent to 10% cocaine was historically used to check for sympathetic pupil denervation.11 However, recent research suggests a more readily available product, apraclonidine, is a reliable (87% sensitivity) and practical solution.13,14
Apraclonidine is an ocular hypotensive agent that acts as a weak alpha-1 agonist and a strong alpha-2 agonist.15 In Horner’s syndrome, upregulation of alpha-1 receptors increases apraclonidine sensitivity and causes denervation and super-sensitivity of the iris dilator muscle. This results in pupillary dilation and lid elevation on the abnormal side, with little to no effect—or even mild miosis on the normal side due to the alpha-2 activity. The apraclonidine test is considered positive if it reverses anisocoria after bilateral instillation.
Twenty-four to 48 hours after applying 1% apraclonidine, the 1% hydroxyamphetamine test may further distinguish a postganglionic or third-order neuron lesion from central or preganglionic causes. Hydroxyamphetamine stimulates the release of stored endogenous norepinephrine from the postganglionic axon terminals into the neuromuscular junction at the iris dilator muscle.
Hydroxyamphetamine drops instilled into a Horner’s syndrome patient’s eye who has intact postganglionic fibers (i.e., first- or second-order neuron lesions) dilate the affected pupil to an equal or greater extent than they do the normal pupil. Therefore, the degree of anisocoria remains the same or decreases. However, hydroxyamphetamine drops instilled into an eye with Horner’s syndrome with damaged postganglionic fibers (third-order neuron lesions) do not dilate the affected pupil as well as they do a normal pupil. Therefore, the anisocoria is likely to increase.
Pharmacological Testing vs. Stat Imaging
Now, apply these pearls to our 38-year-old construction worker. You could certainly do some more investigation into his general health, as well as surgical and recreational history. Does he smoke? Does he have a history of cluster headaches? With that said, and given what we know, this is a case where stat imaging takes precedence over trying to schedule pharmacological testing to localize the lesion—pain trumps pupil testing in this case.
While scheduling, communicate with the radiologist who will be running the tests. Radiologists can often act as a partner in designing an imaging plan based on the clinical insight you provide.
Take the time to educate your staff on the importance of bringing all unilateral ptosis patients to your attention before they are dilated. Horner’s syndrome is not something you want to miss.
1. Walton K, Buono L. Horner syndrome. Curr Opin Ophthal 2003;14:357–63.2. Yanoff M, Duker J. Ophthalmology, 2nd ed. St. Louis, MO: Mosby;2004:1363–9.
3. Flaherty P, Flynn J. Horner Syndrome Due To Carotid Dissection. J Emerg Med. 2011;41:43-6.
4. Borgman C. Horner syndrome secondary to internal carotid artery dissection after a short-distance endurance run: A case study and review. J Optom. 2012;05:209-16.
5. Biousse V, Touboul P, D’Anglejan-Chatillon J, et al. Ophthalmologic manifestations of internal carotid artery dissection. Am J Ophthalmol. 1998 Oct;126(4):565-77.
6. Lee W, Jensen E. Bilateral internal carotid artery dissection due to trivial trauma. J Emerg Med. 2000;19:35-41.
7. Schievink W. Spontaneous dissection of the carotid and vertebral arteries. N Engl J Med. 2001;344:898-906.
8. Parissis H, Young V. Treatment of pancoast tumors from the surgeons prospective: reappraisal of the anterior-manubrial sternal approach. Journal Of Cardiothoracic Surgery. 2010 Nov;10. Available at www.cardiothoracicsurgery.org/content/5/1/102/abstract.
9. Archie V, Thomas C. Superior sulcus tumors: a mini-review. The Oncologist. 2004;9(5):550-5.
10. Kim P, Hains F, Wallace M, Mior S. Pancoast tumour: a case report. Journal Of The Canadian Chiropractic Association [serial on the Internet]. 1993 Dec;37(4):214.
11. Mughal M, Longmuir R. Current pharmacologic testing for Horner syndrome. Curr Neurol Neurosci Rep. 2009;9:384-9.
13. Koc F, Kavuncu S, Kansu T, et al. The sensitivity and specificity of 0.5% apraclonidine in the diagnosis of oculosympathetic paresis. Br J Ophthalmol. 2005 Nov;89(11):1442-4.
14. Freedman K, Brown S. Topical apraclonidine in the diagnosis of suspected Horner syndrome. J Neuroophthalmol. 2005 June;25(2):83-5.
15. Morales J, Brown S, Abdul-Rahim A, Crosson C. Ocular effects of apraclonidine in Horner syndrome. Arch Ophthalmol. 2000 Jul;118(7):951-4.

