Despite the widespread prevalence of herpes virus and adenovirus, their diagnosis can sometimes be deceptive and their treatment can be tricky. This article covers the basics on these vexing viruses, as well as their diagnosis and treatment—and a discussion of the do’s and don’ts of using steroids.
Viral Conjunctivitis Basics
• HSV keratitis. Keratitis caused by herpes simplex virus (HSV) is the most common cause of cornea-derived blindness in developed nations.1 There are approximately 20,000 new HSV keratitis cases and 48,000 recurrences reported annually in the United States, with a national prevalence of 400,000.2
Humans are the only natural host of herpes. These viruses are ubiquitous—in most parts of the world human exposure to HSV-1 is almost universal by late adulthood with a high percentage of exposure occurring in childhood.1
 |
| Fig. 1. Rose bengal stain of herpes simplex ulcer shows peripheral infected cells which pick up greater stain. |
HSVs have an affinity for the sensory ganglion cells and are referred to as “neurotropic” viruses. HSV is commonly divided into Types I and II; Type I typically occurs in the orolabial area and Type II typically in the genital area. Recent studies suggest that this difference is decreasing with time.3
The source of infection is typically through direct contact with infected lesions, salivary droplets from children or adults with active disease (i.e., cold sores), or from asymptomatic virus-shedding carriers.4
Iatrogenic sources of patient infection occur from doctors’ unwashed hands and contaminated applanation tonometer heads. HSV is viable up to two hours on a dry tonometer head and eight hours on one that is moist.5 Swabbing the tonometer head with 70% isopropyl alcohol is almost 100% effective at killing the virus; this should be done between patients along with hand washing with a soapy solution.6
• Adenoviral conjunctivitis. Adenoviral infections are the most common cause for red eye visits to the doctor’s office. Approximately 70% of all cases of acute conjunctivitis present to either primary care or urgent care centers. Optometry and ophthalmology practices see only about 20% of these types of cases.7
The spectrum of disease severity varies with the serotypes: 1 to 11 and 19 are commonly restricted to the eye and cause follicular conjunctivitis, whereas serotypes 3 to 5 and 7 cause pharyngoconjunctival fever. Serotypes 8, 9 and 37 commonly cause the classic epidemic keratoconjunctivitis (EKC).8
Adenoviral infections consist of a biphasic process during which the infectious phase is followed by a variable inflammatory phase. The inflammatory phase tends to begin approximately seven to 10 days post-infection. In the later inflammatory phase, it becomes harder to halt the viral production and shedding.9,10 The virus can be shed for approximately two to three weeks following the infection.1
Adenovirus is a hardy and easily transmissible virus, spreading commonly through hand to eye contact, respiratory droplets and contact with contaminated surfaces or persons involved in primary eye care with other patients.10 Intrafamilial attack rates may vary between 10% to 50%.10
Diagnosis of Viral Eye Disease
The diagnosis of HSV and adenoviral infections manifest in patients with early biomicroscopic signs in the conjunctiva.
Do’s and Don’ts of Using Steroids for Viral Eye InfectionsSteroids have been known to prolong the persistence of viral infection in the cornea, enhance adenoviral replication, prolong viral shedding and lead to a long lasting dry eye.17,18Then again, steroids are often used for comfort and symptomatic ocular relief by addressing pain and suppressing conjunctival and corneal inflammation. Here’s how to use steroids effectively and safely for viral presentations: Because adenoviral conjunctivitis and keratoconjunctivitis are self-limiting entities, sup- portive therapy is a reasonable approach. One study showed 36% of surveyed practitio- ners will always use corticosteroid drops while others never use them. The remaining doc- tors will use them under certain criteria: central infiltrates that decrease vision, intractable pain or the presence of a pseudomembrane.19 A judicious case-by-case approach seems warranted. Many patients with only moderate discomfort appreciate the relief of steroids. If inflammation persists for four or more weeks on the corticosteroids, then their use may be tapered off with the admonition that the disease will “just have to run its course.” In a patient who is still contagious, the use of topical corticosteroids can inhibit cell shedding, leading to a prolonged clinical course.20,21 Research suggests that normal adenovirus clearance is inhibited by the strong anti-inflammatory and anti-immune effects of the corticosteroid. Do’s and Don’ts of Using Steroids for Viral Eye Infections Steroids are typically avoided with herpetic epithelial keratoconjunctivitis or with an epitheliopathy suggestive of Acanthamoeba. The clinical signs of this infection are subtle, although the symptoms of discomfort are more severe and out of proportion. Acanthamoeba keratitis is slow growing and if steroids are mistakenly used, they can be discontinued before extensive damage has been done, as long as the clinician is aware of the possibility. The severe cases of Acanthamoeba have usually occurred after prolonged treatment and where the possibility of this organism was discounted or not considered. Finally, steroids should not be used with presumed fungal keratoconjunctivitis, although these are rare and the clinical picture is usually unambiguous. Side effects of corticosteroids include cataracts and glaucoma. The former problem may take many months to occur; however, as the use of the drops continues in middle- aged or older patients, the clinician runs the risk that a normal age-related cataract may be mistaken for an iatrogenic cataract. Corticosteroid use for longer than four to six weeks is unwise. Usually, the discomfort during the chronic phase is not so severe as to need steroid drops. A steroid-related intraocular pressure rise usually takes weeks and can be easily monitored.16 Cessation of the medication almost always results in pressure returning to baseline. Four to six weeks of drops may be used in most situations, with the steroids then tapered and pressure-reducing drops used if the pressure elevation becomes a concern. |
• HSV diagnosis. HSV keratitis patients commonly complain of a recent onset foreign body sensation with associated photophobia, variable pain/irritation, burning, lacrimation and ocular hyperemia. Questioning the patient about recurrent eye infections is helpful when differentiating HSV early on from other acute red eyes.
Herpetic keratitis usually manifests only in one eye, but bilateral infection can happen. This should bring into question whether the patient is immunocompromised or whether the diagnosis of HSV is the underlying disorder.11
Clinical signs may include dermal eruptions of vesicles around the orolabial area. Ipsilateral preauricular involvement is also common with HSV keratitis. Biomicroscope examination of conjunctiva usually reveals acute or recurrent follicular conjunctivitis, with a pseudomembrane rare in most cases. Dendritic, dendrogeographic or geographic ulcerations on the cornea epithelium are caused by live HSV replication in these cells. The disease may initially appear as a punctate keratitis evolving later into the classic dendrite form.
Use of the AdenoPlus point-of-care test (Rapid Pathogen Screening) may be effective in the punctate stage of HSV keratitis to help confirm the lack of an adenoviral etiology or negative test result.
Standard rose bengal staining will highlight not only the areas of epithelial absence but also the swollen infected epithelial cells surrounding the ulcerated area. (Figure 1) Conversely, the center of the lesion is devoid of cells and stains with sodium fluorescein, while fluorescein may negatively stain the surrounding damaged epithelium. (Figure 2) Under the dendrite, it is not uncommon for the clinician to note a faint stromal infiltrate in the shape of the epithelial lesion. The anterior chamber and corneal endothelium show varying degrees of cellular activity and type.
• Adenoviral diagnosis. Adenoviral ocular disease presents with marked foreign body sensation, photophobia, lacrimation to varying degrees and burning sensation. This occurs in the infected eye with the second eye becoming involved, usually less severely, after the first eye.
Further investigation may reveal an association with outside individuals (family/coworkers) with acute red eyes as significant for patients with EKC—as would be a recent upper respiratory infection or exposure to a community pool or spa in a young/adolescent patient with pharyngoconjunctival fever (PCF).
Clinical findings with the biomicroscope include hyperemia of the lids, caruncle and plica with follicles (Figure 3) mainly located in the inferior palpebral conjunctiva. Pseudomembranes (Figure 4) can be present in one-third to one-half of the cases making the patient more symptomatic. Subconjunctival hemorrhages are common and may obscure the examiner’s view of the follicles in some cases.
The cornea stains with sodium fluorescein and rose bengal to reveal a diffuse, fine, punctate type of stain but without evidence of dendrites or dendritic variations.
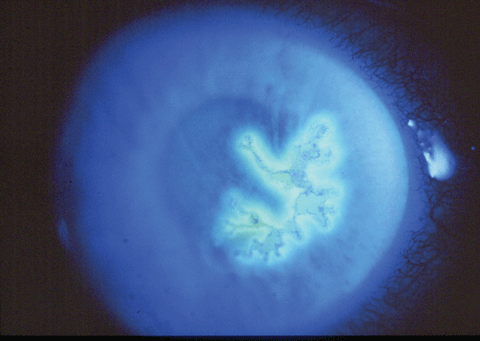 |
| Fig. 2. Herpes simplex dendritic ulcer with sodium fluorescein stain shows a central absence of epithelial cells. Click image to enlarge. |
AdenoPlus testing is most helpful in these cases as it makes for efficient, specific and sensitive diagnosis of an adenoviral infection. It can easily be performed in-office without special equipment. The test targets the hexon protein, which allows the detection of all of the adenoviral serotypes. The test has a reported sensitivity of approximately 89% and specificity of 94%.12 Making a rapid assessment of the viral etiology (HSV vs. adenoviral) can help establish treatment plans to more quickly eradicate the infection or reduce the symptomology of the patient.
Treatment for Adenoviral Infection
When a presumptive diagnosis of adenoviral conjunctivitis is made, topical antibiotics are commonly but improperly prescribed. This is done even though more than half of the cases are viral.13 Antibiotics are prescribed often as a result of:
• Misdiagnosis—the patient actually has a bacterial infection.
• Superinfection—the patient has both types of microorganisms.
• Patient comfort—the drops themselves are often soothing and additionally dilute out the organisms and toxins.
• Patient expectations—the patient seeks therapy that has a reasonable chance of working.
While it may not be the patient’s top concern, the trend in medicine is to avoid topical antibiotics absent the certainty of a bacterial infection because of the public health risk of antibiotic microbial resistance.14,15 They are ineffective against viral conjunctivitis and have little therapeutic impact. They may delay the proper treatment and can cause antibiotic resistance, toxicity and allergy.
Currently, no approved topical antiviral drug exists for adenoviral conjunctivitis. Treatment is typically supported with the use of cool compresses and/or artificial tears for comfort several times a day. Cycloplegic agents may be used for severe photophobia.
Because the virus is so contagious, prevention of its spread to family, coworkers and fellow students is a must. Mainstays are frequent handwashing and avoidance of touching the infected eye, as well as sanitizing instruments and surfaces in the eye care office. During active infection, recommend patients (whether students or workers) to stay at home to avoid infecting others.
Zirgan (ganciclovir 0.15%, Bausch + Lomb) has been investigated as a topical treatment for adenoviral keratoconjunctivitis. In one study, topical Zirgan resulted in a quicker cessation of signs and symptoms; however, this was not statistically significant.14
For patients with acute, painful adenoviral infection, consider a one-time instillation of Betadine 5% solution. (See “Can Betadine Blast Out Acute Viral Conjunctivitis?” page 86.) Pseudomembranes can be manually peeled in office every few days.
Treatment for Herpes Virus Infection
Treatment for herpetic eye disease depends on the presentation and the location of the infection, and may include oral antivirals, topical antivirals and topical corticosteroids. Here are the recommended treatments for the following forms of ocular herpetic infection:
• Herpes zoster ophthalmicus (shingles). Initial treatment of herpes zoster ophthalmicus is almost always with an oral antiviral agent so as to lessen the severity and duration of the dermatologic disease. The least expensive treatment also requires the most doses: Zovirax (acyclovir, GlaxoSmithKline) 800-1,200mg PO five times per day. Other treatments are Valtrex (valacyclovir, GlaxoSmithKline) 1,000mg PO TID or Famvir (famciclovir, Novartis) 500mg TID. All are prescribed for seven to 10 days for patients with good renal function.
Without treatment, and usually even with it, skin lesions will erupt in the dermatome of the affected nerve(s). Early treatment lessens the severity and duration of the infection. The keratitis that can occur with zoster is usually much milder than that with simplex. The micro dendrites or pseudodendrites that occur are generally not a significant problem beyond irritation to the patient. They often have overlying mucus and may form mucus filaments or plaques.
Bacterial superinfection is usually prevented with an antibiotic such as Vigamox (moxifloxacin 0.5%, Alcon) TID. If there is a secondary sterile anterior chamber reaction, a cycloplegic such as Atropen (atropine 1%, Meridian) or Cyclogyl (cyclopentolate 1%, Alcon) may be used several times a day. Pred Forte (prednisolone acetate 1%, Allergan) may be used QID for secondary iritis and photophobia. The use of topical steroids for zoster are not contraindicated as they are for simplex. (See “Do’s and Don’ts of Using Steroids for Viral Eye Infections,” page 79.) Microdendrites/pseudodendrites have been shown to respond to Zirgan five times per day.15
A small minority of cases of zoster keratitis can lead to persistent keratitis, progressing into the stroma without a subsequent epithelial defect (disciform keratitis) or with absence of epithelium (necrotizing keratitis). Supportive therapy with steroids and cycloplegics is administered with the former case. With necrotizing keratitis, conjunctival flaps or even corneal transplants must be done (though this is uncommon in most cases). Intraocular inflammation may persist leading to glaucoma, cataracts and anterior or posterior synechiae and treatment must be directed at these problems.
• Herpes simplex keratitis. Early herpes simplex ocular infection may not initially show a dendrite. Occasionally the dendrite may present on the conjunctiva. A search with fluorescein or another stain (rose bengal or lissamine green) must be done to ensure detection of conjunctival epithelial defect in a moderately red eye. If the conjunctivitis is unilateral, then the treatment for bacterial conjunctivitis may be implemented while acknowledging the possibility of early presentation HSV. Steroids are contraindicated. If no dendrite appears in four to seven days, then steroids could be used in what is more likely bacterial blepharoconjunctivitis. If the patient develops dendrites or initially presents with them, then antivirals are indicated.
For more than 50 years, topical antiherpetic medications have been the first line of treatment. The mainstay of antiviral medication has been Viroptic (trifluridine 1%, Monarch) used usually nine times per day on initial presentation. As the dendrites begin to heal, the medication is tapered to five times per day and then discontinued. Because toxicity may occur, treatment is rarely continued longer than two to three weeks—except with a corneal transplant patient in need of prolonged treatment.
Recently, Zirgan five times per day has been used to treat epithelial keratitis; this can be used as a primary treatment or when the patient is resistant to Viroptic. The go-to topical ointment Vira-A (vidarabine, Abcam) is no longer available. Initial therapy now often consists of oral treatment: acyclovir 400mg five times per day, valacyclovir 500mg BID or famciclovir 250mg BID—any of which are to be taken for seven to 10 days.
Orals aren’t often used as first line therapy for HSV except when:
1. There is a high risk of corneal toxicity.
2. The patient is immunocompromised.
3. The patient is a child in whom it is difficult to instill drops.
Treatment of superficial epithelial corneal disease occurs because of supratherapeutic levels of the antiviral in the precorneal tear film. Failure of epithelial lesions to resolve after two weeks suggests either epithelial toxicity, neurotrophic keratopathy or drug resistance.
An additional treatment at the initial presentation of a patient with HSV dendrites is to perform manual epithelial debridement with a cotton-tipped applicator. Removing infected and injured epithelial cells decreases the viral load on the cornea and can hasten the healing process.
• HSV geographic ulcer. Geographic ulcers are dendrites that widen and spread out while still remaining largely epithelial. The topical dose of either Viroptic or Zirgan is unchanged. If oral therapy is chosen, the dosage is increased to that for the treatment of herpes zoster: acyclovir 800mg five times per day, valacyclovir 1,000mg TID or famciclovir 500mg BID.
As with all purely epithelial herpes infections, avoid steroids unless there is a good reason, such as the presence of a corneal graft. Be aware that when epithelial herpes is severe enough, as in a geographic ulcer, the doctor may imagine that there is stromal involvement when there is not. Bowman’s membrane can become hazy, but the disease is still limited to the epithelium.
• HSV disciform (stromal) keratitis. In this case, the stroma is swollen, usually in the central cornea. The stroma appears hazy and/or Descemet’s membrane is wrinkled, indicating thickening. The overlying epithelium is intact and dendrites have usually resolved.
In this case—usually a sterile autoimmune process—steroids are added at four to eight times per day while maintaining topical Viroptic coverage five times per day, Zirgan TID, or oral antivirals given in the lower-dose regimen, such as with dendritic keratitis.
With prolonged treatment, more than three weeks of topical Viroptic may lead to ocular surface disease. Treatment is then switched to prophylactic doses of oral agents such as acyclovir 400mg BID while maintaining the steroid drops and monitoring the intraocular pressure.16
• Necrotizing (stromal with epithelial defect) keratitis. Here, the epithelium is gone and there is a stromal ulcer. Because of the infectious inflammation and the viral neurotropic nerve damage, it is difficult to induce the epithelium to cover and stabilize the dehydrating and thinning surface.
There is insufficient clinical trial data to support type and length of treatment. Steroids are often used, albeit cautiously. Pred Forte is used BID along with topical or oral antivirals. these agents are gaining prevalence due to the lack of toxicity. Oral antivirals are used in the higher doses for seven to 10 days, and the low-dose steroid is tapered. Ocular surface protection with a conjunctival flap or amniotic membrane must be considered. Topical antivirals are usually avoided because epithelial healing is so problematic and topical medications would likely only worsen the healing.
• Endothelial keratitis. Some patients who have a limited anterior chamber reaction, with fine keratic precipitates on the endothelial surface. This inflammatory, primarily autoimmune reaction should be treated with Pred Forte six to eight times per day along with a low-dose oral antivirals. The Pred Forte may then be tapered and the orals reduced to the prophylactic dose.
The eye remains the premier testing ground for many drugs under evaluation in experimental human clinical trials. With the development of more specific and systemic antiviral agents and the multiple viral infections manifested in ocular disease, the prospects for new and effective therapies remain highly promising in the near and distant future.
Dr. Sendrowski is a professor at the Southern California College of Optometry, and the Chief of the Ophthalmology Consultation and Special Testing Service at the Marshall B. Ketchum University Eye Center, in Fullerton, Calif.
Dr. Maher is a general ophthalmologist with fellowship training in cornea and external disease in private practice in Torrance, Calif.
Thanks to Jeremy-Ann Ham of the Department of Molecular Cell Biology and Physiology at California State University at Long Beach for assistance with this paper.
|
1. Knickelbein JE, Hendricks RL, Charukamnoetkanck P. Management of stromal keratitis: an evidence based review. Sur Ophthal 2009;54(2):226-34. 2. Paving-Langston D. Viral disease of the ocular anterior segment: basic science and clinical disease. In Foster S, Azar D, Dohlman C. eds. Philadelphia, PA: Lippencott, Williams & Wilkins 2005;297-397. 3. Pepose JS, Keadle TL, Morrison LA. Ocular herpes simplex changing epidemiology, emerging disease patterns, and the potential of vaccine prevention and therapy. Am J Ophthalmol 2006;141:547-57. 4. Remejier L, Osterhaus A, Verjans G. Human herpes simplex virus keratitis: the pathogenesis revisited. Ocul Immunol/Inflamm 2004;12:255-85. 5. Liesegang T. Ocular Virology. In: Albert D, Jacobies F. eds. Philadelphia, PA: WB Saunders 2000;171-98. 6. Rutala W, Weber D, Healthcare Infection Control Practices Advisory Committee (HICPAC). Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008. CDC. Available at www.cdc.gov/hicpac/pdf/guidelines/Disinfection_Nov_2008.pdf 7. Young RC, Hodge DO, Liesgang TJ, et al. Incidence, recurrence, and outcomes of herpes virus simplex virus eye disease in Olmstead County, Minnesota, 1976-2007. The effect of oral antiviral prophylaxis. Arch Ophthalmol 2010;128(9):1178-83. 8. Ghebremedhin B. Human adenovirus: Viral pathogen with increasing importance. Eur J Microbiol Immunol (Bp). 2014;4(1):26–33 9. Liebowitz HW, Pratt MV, Flagstad IJ et al. Human conjunctivitis. I. Diagnostic evaluation. Arch Ophthalmol 1976;94:1747-9. 10. Azar MB, Dhaliwal DK, Bower KS. Possible consequences of shaking hands with your patients with epidemic keratoconjunctivitis. Am J Ophthal 1996;121:711-12. 11. Wilhelmus KR, Falcon MG, Jones BR. Bilateral herpetic keratitis. Br J Ophthalmol 1981;65:385-9. 12. Sambursky R, Tucker S, Schirra F, et al. The RPS adeno detector for diagnosing adenoviral conjunctivitis. Ophthalmology 2006;113:1758-64. 13. Yabiku ST1, Yabiku MM, Bottós KM, Araújo AL, Freitas Dd, Belfort Jr R. Ganciclovir 0.15% ophthalmic gel in the treatment of adenovirus keratoconjunctivitis. Arq Bras Oftalmol 2011;74(6):417-21. (Article in Portuguese) 14. Everett HA, Little PS, Smith PW. A randomized controlled trial of management strategies for active infective conjunctivitis in general practice. BMJ. 2006;333(7563):321. 15. Aggarwal S, Cavalcanti BM, Pavan-Langston D. Treatment of pseudodendrites in herpes zoster ophthalmicus with topical ganciclovir 0.15% gel. Cornea. 2014; 33(2):109-13. 16. Kowalski RP, Foulks GN, Gordon YJ. An overview comparing treatment regimens for ocular infections: community versus academia. Ann Ophthalmol. 2000;32:295-300. 17. Melton R, Thomas R. Current trends in medical management. American Academy of Optometry. Seattle, WA 2013; www.eyeupdate.com. 18. Shorlin JP. What’s the buzz about betadine? Review of Optometry. 2011 Sept 15. 19. Butt AH, Chodosh J. Adenoviral keratoconjunctivitis in a tertiary care eye clinic. Cornea. 2006;25:199-202. 20. Romanowski EG, Yates KA, Gordon YJ. Topical corticosteroids of limited potency promote adenovirus replication the Ad5/NZW rabbit ocular model. Cornea. 2002;21:289-91. 21. Becker B, Mills DW. Corticosteroids and intraocular pressure. Arch Ophthalmol. 1963;70:482-91. 22. Pihos AM. Epidemic keratoconjunctivitis: a review of current concepts in management. J Optom. 2013;6(2):69-74. |

