 |
A 72-year-old Caucasian female presented as an emergency office visit with complaints of intermittent, horizontal diplopia and ptosis of both eyes that worsened when she was fatigued. She also described increased difficulty standing up when sitting. Additional history taking revealed increased arthritic-like pain, increased fatigue and variable lower limb weakness.
Her medical history was remarkable for hypothyroidism, rheumatoid arthritis, hypercholesterolemia, Sjögren’s syndrome and post-herpetic neuralgia. Surgical history included spinal fusion (L3, L4). Her ocular history was remarkable for keratoconjunctivitis sicca, corneal erosion, cataract and vitreomacular traction syndrome in the left eye.
Evaluation
Best-corrected visual acuity was 20/50 OD and 20/60 OS, attributed to bilateral cataracts. Pupils were equal, round and reactive to light. Intraocular pressures were normal. Given the presentation of progressive, fatigable upper lid ptosis, non-comitant abduction deficits, and normal pupils, myasthenia gravis (MG) was the diagnostic concern. We ordered an ice test to evaluate for improvement of the ptosis and diplopia. The ptosis improved bilaterally while the diplopia remained unchanged.
Taking into account her complaints of lower limb weakness, variable ptosis and diplopia, we scheduled an appointment for consultation with a neurologist for electromyography (EMG) to assess the nerve-muscle conduction of her lower limbs and confirm systemic involvement.
EMG revealed an increment of compound muscle potential (CMAP) amplitude of more than 100% on repetitive nerve stimulation, which is not expected in MG but is most consistent with Lambert-Eaton myasthenic syndrome (LEMS). Laboratory testing came back negative for all antibodies related to MG. Subsequently, additional labs were ordered to investigate for positive calcium-channel antibodies associated with LEMS. Because of the presence of positive calcium channel antibodies and CMAP amplitude of more than 100% on repetitive nerve stimulation, a diagnosis of LEMS was made.
Subsequently, the patient was prescribed 100mg of Mestinon (pyridostigmine, Bausch + Lomb). At her two-week follow-up, there was complete resolution of the diplopia and partial improvement of the ptosis bilaterally. She is currently being managed on 100mg of Mestinon.
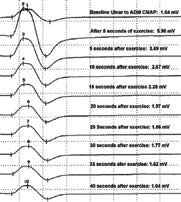 |
| EMG shows initial increment and gradual decline of compound-muscle action potentials following sustained exercise, characteristic of LEMS. |
Discussion
Lambert-Eaton myasthenic syndrome is a rare autoimmune disease affecting possibly as few as 400 people across the United States. It is caused by autoantibodies against voltage-gated calcium channels in the neuromuscular junction seen as a paraneoplastic syndrome in the setting of small cell lung cancer (SCLC), as well as being idiopathic.1,2,3 LEMS has a clinical triad of proximal muscle weakness, autonomic symptoms and reduced tendon reflexes; it is also associated with small cell lung cancer in approximately 60% of cases.2
LEMS patients may present with symptoms that are quite similar to those with MG, given the similarities in pathophysiology.2 Additionally, patients with either form of LEMS or MG are more likely to have additional concomitant autoimmune diseases, as was observed in our patient.4,5 LEMS is diagnosed after the assessment of clinical symptoms and signs, antibody testing and electrophysiological testing.
LEMS patients test positive for voltage-gated calcium channel (VGCC) antibodies 85 to 90% of the time. EMG testing will show low CMAP that depresses even further when tested at low frequencies—LEMS patients show decreased CMAP amplitudes 94 to 98% of the time.6 Post-exercise stimulation will show increased CMAP more than 100%, which is considered abnormal and is 100% specific for LEMS, with a sensitivity of 84 to 96%.6
The diagnosis of LEMS may be delayed because it has a non-specific pattern of atrophy that may appear similar to other neurological diseases, particularly in those patients with idiopathic LEMS.3 In some cases, LEMS may be confused with Guillain-Barre syndrome or amyotrophic lateral sclerosis (ALS).3 The most common mimic of LEMS, however, is MG. Initial presenting symptoms of MG include ptosis and diplopia, where LEMS most often starts with mild upper leg weakness.3
As previously mentioned, there are rare cases (5%) of LEMS that present initially with ocular symptoms in contrast to the 90% of cases of myasthenia gravis that present initially with ocular symptoms.7 LEMS is virtually excluded in patients who present with ocular weakness first, which can ultimately delay proper diagnosis and additional testing to rule out SCLC commonly associated with LEMS.3,7 Some patients with MG have an initial presentation of limb weakness, but their weakness is typically of the arms (as opposed to the legs in LEMS) and proceeds from head to feet, which is opposite of what is observed in LEMS.7
While practitioners see several clinical similarities between myasthenia gravis and LEMS, the two diseases are serologically different. For LEMS patients, 85% to 90% test positive for VGCC autoantibodies, where 50% to 70% of ocular MG patients test positive for anti-acetylcholine receptor antibodies.8,9 In patients with systemic MG, up to 90% will test positive for the anti-acetylcholine receptor antibodies.9
Treatment
While several diseases may mimic LEMS, it is important to obtain a proper diagnosis so that the correct treatment may be initiated. The treatment of choice for LEMS is 3,4-diaminopyridine (amifampridine).10,11 This drug can improve muscle strength in patients with LEMS and is generally well tolerated with few side effects.10 It blocks the efflux of potassium ions on the presynaptic nerve to prolong depolarization, keeping the calcium channels open for longer and allowing for increased release of acetylcholine.10
Side effects may include digital paresthesias or gastrointestinal symptoms, while some serious side effects included seizures and tachycardia with higher dosages of the drug.10 Amifampridine blocks VGCCs, which elongates the action potentials at the motor nerve terminals, allowing for increased activity at the neuromuscular junction.12 In cases where amifampridine is unavailable, pyridostigmine may be used.3
Amifampridine, under the name Firdapse (Catalyst), is the first FDA-approved treatment for LEMS in the United States, and our patient was very eager to talk with her neurologist about switching from her pyridostigmine treatment to amifampridine.13 The reason she was not initially started on Firdapse was due to the limited clinical use by the neurologist.
When managing these patients, it is important to realize that, despite treatment, many patients still exhibit bothersome diplopia and or ptosis. Supportive measures can include temporary prism or occlusion therapy for patients with persistent diplopia and ptosis. Surgery can be considered in patients whose symptoms are stable for a minimum of six months.
While rare, LEMS is still an important differential to keep in mind for patients who experience variable ptosis and diplopia. LEMS is most commonly seen as a paraneoplastic disorder and while it was determined that this patient had idiopathic LEMS, all patients must undergo CT of the lung to discount SCLC. It is for this reason that LEMS should be considered in the differential diagnosis of a patient with variable ptosis, diplopia and proximal muscle weakness.
1. Parsons K. Lambert-Eaton Myasthenic Syndrome. In: NORD Guide to Rare Disorders. Philadelphia, PA: Lippincott Wiliams & Wilkins; 2003:621. 2. Mareska M, Gutmann L. Lambert-Eaton myasthenic syndrome. Semin Neurol. 2004;24(2):149-153. 3. Titulaer MJ, Lang, Verschuuren JJ. Lambert-Eaton myasthenic syndrome: from clinical characteristics to therapeutic strategies. Lancet Neurol. 2011;10(12):1098-1107. 4. Wirtz P, Bradshaw J, Wintzen AR, Verschuuren JJ. Associated autoimmune diseases in patients with the Lambert-Eaton myasthenic syndrome and their families. J Neurol. 2004;251(10):1255-9. 5. Nacu A, Andersen JB, Lisnic V, et al. Complicating autoimmune diseases in myasthenia gravis: a review. Autoimmunity. 2015;48(6):362-8. 6. Oh SJ, Kurokawa K, Claussen GC, Ryan HF. Electrophysiological diagnostic criteria of Lambert-Eaton myasthenic syndrome. Muscle Nerve. 2005;32:515-20. 7. Wirtz PW, Sotodeh M, Nijnuis M, et al. Difference in distribution of muscle weakness between myasthenia gravis and the Lambert-Eaton myasthenic syndrome. J Neurol Neurosurg Psychiatry. 2002;73(6):766-8. 8. Lennon V, Kryzer TJ, Griesmann GE, et al. Calcium-channel antibodies in the Lambert-Eaton syndrome and other paraneoplastic syndromes. N Engl J Med. 1995;322(22):1467-74. 9. Benatar M. A systematic review of diagnostic studies in myasthenia gravis. Neuromuscul Disord. 2006:16(7):459-67. 10. Keogh M, Sedehizadeh S, Maddison P. Treatment for Lambert-Eaton myasthenic syndrome. Cochrane Database Syst Rev. 2011;(2):CD003279. 11. Skeie G, Apostolski S, Evoli A, et al. Guidelines for treatment of autoimmune neuromuscular transmission disorders. Eur J of Neurol. 2010;17(7):893-902. 12. Wu ZZ, Li DP, Chen SR, Pan HL. Aminopyridines potentiate synaptic and neuromuscular transmission by targeting the voltage-activated calcium channel ß subunit. J Biol Chem. 2009;284(52):36453-61. 13. FDA approves first treatment for Lambert-Eaton myasthenic syndrome, a rare autoimmune disorder [press release]. Silver Spring, MD. United States Food and Drug Administration. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm627093.htm. November 28, 2018. Accessed February 8, 2019. |

