Modern scientific methodology—specifically, the randomized clinical trial (RCT)—is considered a critical component of evidence-based medicine to separate fact from fiction regarding interventions, drugs, nutrition and other strategies we use to treat our patients safely and effectively. A well-designed, large-scale clinical trial that is considered valid, reliable and unchallenged is critical to the emergence of a standard of care; however, the results achieved are only as good as the design of the clinical trial.
New research concerning AMD has raised a few eyebrows among enthusiasts of ocular nutrition. The dialogue centers on two particular studies—the AREDS-2, authored by National Eye Institute Deputy Clinical Director Emily Chew, MD, and a polymorphism genomic paper, authored by Carl Awh, MD, a private practice ophthalmologist in Nashville. Dr. Chew’s research is a classic RCT that investigated the addition of new nutrients to the current AREDS formula, while Dr. Awh’s study predicted subject response to AMD with antioxidants and zinc.
Although both studies used the same clinical information, the genomic paper reanalyzed a statistically significant sample of subjects’ data and genetic samples from AREDS-1. The methodologies differed significantly, which led to contrasting conclusions in treatment. The resulting controversy places us in a precarious position as we determine appropriate nutrient recommendations for patients with moderate or advanced AMD and those at risk for AMD.
At times, clinical trials are challenged due to confounding errors, such as investigator bias, inaccurate assumptions about the drug or nutrient interaction, or an insufficient number of subjects enrolled in subsets, causing statistically insignificant findings in the analysis of primary or secondary randomization.1
 AREDS-1
AREDS-1
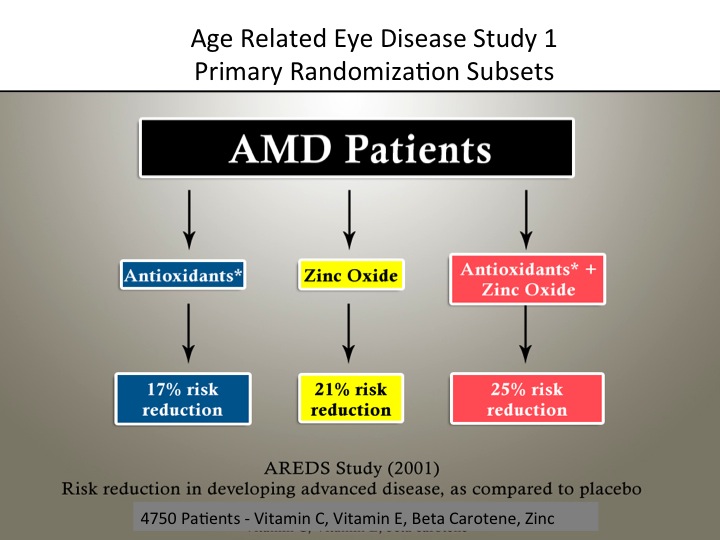
Widespread public use of commercial vitamins and minerals to treat AMD—with few definitive answers on their safety and efficacy—initiated the AREDS-1 RCT in 2005. The National Eye Institute (NEI) sponsored this randomized, placebo-controlled, double-masked clinical trial primarily concerned with studying the effects of a nutrient combination on the progression of AMD.
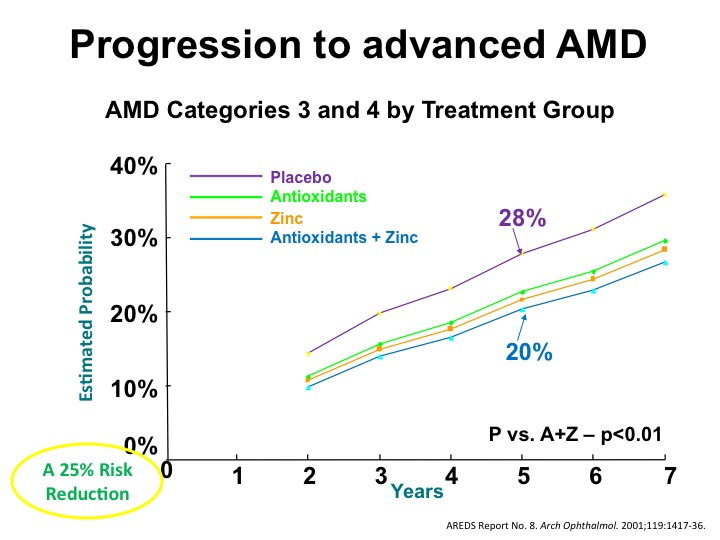
According to the AREDS-1 findings, the risk to advanced AMD decreased by 25% in stage III. Though the formulation slowed the progression from stage III to stage IV, it did not halt the progression of AMD (figure 1).1 The zinc subset had a 21% risk reduction and antioxidants had a 17% risk reduction related to the progression of AMD in stages III and IV (figure 2).2 The evidence did not support the use of these supplements for prevention in the early stages I and II.
Overall, the results were statistically significant but modest. The Cochrane Reviews, an RCT repository that provides systematic reviews of primary research in human health care and health policy, found that the generalizability of these findings to other populations with different nutritional status was unknown; therefore, long-term harm from supplementation could not be ruled out.3 Beta-carotene, for example, has been found to increase the risk of lung cancer in smokers and vitamin E has been associated with increased heart-failure risk.3, 4
There were also some concerns related to zinc and genitourinary complications. Many clinical investigators questioned the benefits of zinc and the antioxidant supplements used; they also wanted more time to study the effects of higher dose supplements.
Based on the AREDS-1 conclusions, should patients with moderate to severe AMD take antioxidant supplements? Some investigators voiced concerns about how the study was funded, and pointed out that there had been no replication of the data. Others noted the lack of long-term safety information, coupled with only a modest effect in reducing progression of AMD.
Encouraged by the preliminary data from AREDS-1, clinical investigators sought to discover if manipulation of the original formula could achieve double the AREDS-1 effect (a 50% decrease in progression of AMD). Many clinicians wanted to change dosing and include some nutrients not used in AREDS-1, such as the carotenoids lutein and zeaxanthin, which are found in high concentrations in the macula, but were not commercially available at the time of the original study. Further, previous studies associated high levels of lutein and zeaxanthin intake with lower risk of AMD.5-8 These nutrients are believed to augment macular pigment optic density (MPOD), which protects the photoreceptors and the retinal pigment epithelium from the oxidative damage associated with exposure to blue light.
A follow-up analysis of AREDS-1 concerned the dietary habits of subjects who answered a food frequency questionnaire at baseline. The study indicated that subjects who consumed fish with long chain omega-3 fatty acids experienced a 25% decrease in AMD progression.9 In addition, several investigators of independent peer-reviewed studies have reported findings that support omega-3 fatty acids in the management of AMD.9-11
Other effects of omega-3 in the literature suggest the following benefits: decreased triglyceride levels, which may affect atherosclerotic plaque formation; lowered blood pressure; reduction in heart attack risk, stroke and dangerous abnormal heart rhythm in patients with known heart disease; anti-inflammatory effect in patients with rheumatoid arthritis; and reduction in risk of breast and prostate cancers.12-17
Researchers also wanted to know whether reduced levels of zinc would affect the formulation’s performance in decreasing progression of AMD, and if reduced zinc levels would decrease genitourinary complications.
There were concerns that current or prior smokers had a higher risk of lung cancer with beta-carotene in the formulation, so investigators also wanted to test whether eliminating beta-carotene altogether and substituting lutein/zeaxanthin would affect the results.
AREDS-2
Researchers started enrollment for AREDS-2 in 2006 with the intention of studying additional nutrient combinations against the benefits of the existing formula. The primary randomization in AREDS included the original formula plus one of the following:
> 10mg of lutein/2mg of zeaxanthin
> Omega-3 fatty acids: 350mg docosahexaenoic acid (DHA)/650mg of eicosapentaenoic acid (EPA)
> Lutein/zeaxanthin plus DHA-EPA above
> The secondary randomization evaluated three subsets using the original formula plus one of the following:
> No beta-carotene
> Low zinc
> No beta-carotene and low zinc
Interestingly, the addition of lutein and zeaxanthin, and DHA/EPA, or both in the primary analysis, did not further reduce the risk of progression to advanced AMD.17
The authors of AREDS-2 sought to determine if a statistically significant sample of the general population would benefit from a specific nutrient formula. The study provided useful evidence to justify a redesign of the original AREDS-1 formula, since reducing the level of zinc and eliminating the beta-carotene did not reduce the efficacy of the supplements. However, there were confounding factors that could have adversely influenced the results.
AREDS-2 Uncertainties
• Study design issues. There were admitted limitations to the study results due to “a complicated design involving a secondary randomization which may have affected our ability to evaluate the role of lutein and zeaxanthin and DHA+EPA to the AREDS formula.”17
• No control group. Due to the modest success of AREDS-1 results, it was ethically necessary to offer prior subjects of AREDS-1 the choice of taking the original AREDS-1 formula or not. In fact, most subjects opted to continue the original formula. Only 19 of about 4,000 (less than 0.05%) of the total enrolled subjects in AREDS-2 did not take the original AREDS-1 formula. Therefore, there was no true control group to compare findings in the AREDS-2 primary analysis.
• Omega-3 confounding factors. The study showed that taking 1000mg of omega-3 did not affect the progression in AMD patients. Some possible reasons include use of the ethyl ester form of omega-3, which is not as bioavailable as the triglyceride form, and the lipid-lowering effect of 1,000mg of omega-3, which may not be sufficient to obtain a desirable result.
Further, many subjects did not follow the study guidelines regarding their recommended nutrient intake. Approximately 84% of subjects took slightly more than 75% of the supplement pills, which could have skewed the results. Fourteen percent of subjects admitted to taking unauthorized additional supplements, either alone or in combination with their supplement pills, during the trial.
• Non-representative sample. AREDS-2 inducted mainly female, well-nourished whites; thus, the study sample was not representative of the biological diversity of the general population. The study group reported that they already consumed supplements, multivitamins or foods rich in lutein, zeaxanthin and omega fatty acids, in addition to the original AREDS supplement. AREDS-2 clinical investigator Paul Bernstein, MD, PhD, a leading carotenoid researcher, confirmed this.
Dr. Bernstein’s work showed a linear relationship between diet or carotenoid-rich supplements and a more robust MPOD. He performed an ancillary study on his AREDS-2 subjects in which he measured MPOD and found their “usage of lutein and zeaxanthin supplements was exceedingly high, suggesting that we enrolled a very nutritionally aware cohort.”20 The inclusion of a larger group of subjects who have statistically significant nutrient deficiencies would likely offer more practical clinical information than studies on well-nourished subjects.
• Nutrient interactions. The primary study conclusion—that lutein and zeaxanthin with or without omega-3 fatty acids did not further reduce the risk of progression—may have been influenced by carotenoid competition.
 Interestingly, when compared with the subgroup that used the original AREDS formula plus lutein and zeaxanthin (without beta-carotene) to the subgroup in the primary randomization of original AREDS formula containing beta-carotene, lutein and zeaxanthin, there is an 18% lower risk of AMD progression. This result may occur because lutein, zeaxanthin and beta-carotene are carotenoids that do compete for absorption the human body.20 Carotenoid competition may explain, in part, why lutein and zeaxanthin had no overall effect in the primary randomization, but a statistically significant effect when beta-carotene was removed in the secondary randomization. Therefore, this confounding error may be viewed by some observers as a significant study flaw affecting the results of the primary study.
Interestingly, when compared with the subgroup that used the original AREDS formula plus lutein and zeaxanthin (without beta-carotene) to the subgroup in the primary randomization of original AREDS formula containing beta-carotene, lutein and zeaxanthin, there is an 18% lower risk of AMD progression. This result may occur because lutein, zeaxanthin and beta-carotene are carotenoids that do compete for absorption the human body.20 Carotenoid competition may explain, in part, why lutein and zeaxanthin had no overall effect in the primary randomization, but a statistically significant effect when beta-carotene was removed in the secondary randomization. Therefore, this confounding error may be viewed by some observers as a significant study flaw affecting the results of the primary study.
Pharmacogenomics and AMD
In the past, large-scale RCT studies allowed industry to develop “blockbuster” drugs to treat an average population. Many now contend that the future of medical studies may focus on a more personalized approach to medicine—that is, developing interventions, drugs or nutrients to treat subsets of patients, each of whom may respond differently based on individual genetic factors.21
While the RCT is invaluable in understanding whether one treatment is better than another on a general platform, the findings may be harder to apply individually. Research gleaned from a combination of RCT and genetic trials may be on the horizon. Pharmacogenomics—the way genes influence response to nutrients and drugs—is unique and different for each individual.22 As genetic research moves forward, the evolution of personalized recommendations will become more commonplace.
Predicting AMD Pathogenesis
The human genome has been catalogued and genetic variations have been identified as single nucleotide polymorphisms (SNPs), which are considered a measure of genetic similarity used for genotyping or genetic fingerprinting. SNPs associated with AMD are located on genes that affect the biological pathways of the disease, and have been confirmed in the literature. Genes involved with the complement cascade of the immune system have a direct effect on the progression of AMD.23 Thus, AMD has the strongest genetic contribution of all human multi-genetic diseases.23
Additionally, the pathogenesis of AMD may be predicted using a combination of fundus diagnosis and determination of genotype.24 A currently available, validated genetic test (Arctic Dx) may provide greater sophistication in how we view an individual’s future risk of AMD progression.25 Using genotyping, fundus diagnosis and health care history together, an individualized prediction of advanced AMD risk over time can be generated.24
Reanalysis of AREDS-1 Using Genomic Testing
Genetic research has shown that SNPs of human variation complement factor (CFH) and age-related maculopathy sensitivity 2 (ARMS2) each have important roles in AMD.26
Dr. Awh, et al., authored a clinical study that obtained statistically significant AREDS-1 data from subjects—including genetic information contained in saliva samples. In a novel twist of methodologies contrasting RCT and genomic testing, investigators did not obtain average population statistics, as is routine in an RCT like AREDS-1. Rather, the authors sought to compare how the original AREDS formula subsets—zinc, antioxidants, and zinc plus antioxidants—would affect progression to AMD when sorted by genotype that were composed of high-risk genes. Dr. Awh was specifically interested in results concerning one or two risk alleles of CFH or ARMS2.
Dr. Awh’s study analyzed AREDS-1 data for up to 12 years. Subjects with the ARMS2 allele(s) benefited more from the formulation with zinc alone. Furthermore, these subjects had a higher risk of progression to AMD when they also were treated with antioxidants. Subjects with the CFH risk allele(s) benefited more with antioxidants than with the complete AREDS-1. Interestingly, when this group was treated with zinc, there was an associated increase in the risk to progression of AMD. Those with both one ARMS2 and one CFH alleles showed reduced progression using the original AREDS-1 formula of zinc plus antioxidants. Dr. Awh extrapolated the AREDS-1 data to genotype and predicted that the optimal treatment for 49% of the study patients would be a nutrient combination different from the complete AREDS-1 formulation.
The scientific literature includes some studies that support Dr. Awh’s contention that there could be a harmful relationship between CFH and ARMS2 with nutrients in the AREDS-1 formula. A retrospective analysis done in 2008 found that the AREDS supplement may be related to the CFH high-risk SNP.26 In 2009, researchers found “significant interaction between the number of risk alleles for the CFH Y402H variant and treatment, whereby patients with the (high risk) genotype were less likely to benefit from the antioxidant-mineral supplementation than subjects with the TT and CT genotypes.27 In 2012, researchers demonstrated that a strong association exists between all stages of AMD and the ARMS2 SNP.28 Two years earlier, Stephen Perkins and colleagues outlined the biological plausibility of the CFH/zinc interaction.1 This publication points specifically at high-risk CFH alleles and zinc.30
Dr. Awh shared his findings about genotype and progression to AMD using AREDS supplements at the American Academy of Ophthalmology’s annual meeting in late 2013. Afterward, Dr. Chew offered a dissenting view and reported that his data did not complement hers. As there is no consensus among retina specialists on the matter, a comprehensive scientific review is needed to reconcile the conflicting data.
AREDS-1 was the first glimpse into a nutrient strategy to help combat AMD based on traditional evidence-based science. The extension study, AREDS-2, confirmed that lutein and zeaxanthin are more beneficial and safer than beta-carotene in reducing progression of the disease. Studies on the benefits of omega-3 fatty acids are ongoing, while many practitioners will continue to recommend DHA and EPA in different forms and dosages.
The results of AREDS-2 should be considered against its ethical considerations, including the absence of a control group, design complexity, nutrient interaction, subject sampling issues and the effectiveness of additional nutrients studied. In hindsight, it may have been clinically relevant to analyze more subjects who were less nourished than the individuals reflected in AREDS-2.
The AREDS RCT also opens a new door to clinical genetic trials. Dr. Awh predicated his recent work on a clinically rich time capsule of information from AREDS-1, collected for more than 10 years. Study investigators were able to fast-track more than a decade of data by looking at influences by genotype rather than by an average population.
Certainly, the debate to recommend personalized genetic testing or to use a classical RCT will evolve. Because AMD has the strongest genetic contribution of all human multi-genetic diseases, it seems likely that at least some form of personalized genetic testing will help us better protect our patients from the visual ravages of AMD.
Dr. Ruskin is the president of the College of Optometrists of Ontario, and is in private practice in Toronto. He is the current chair of the Ocular Nutrition special interest group within the American Academy of Optometry. He has no direct financial interest in any of the products mentioned in this article.
1. Titler MG. The Evidence for Evidence-Based Practice Implementation. In: Hughes RG, ed. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Rockville, MD: Agency for Healthcare Research and Quality; 2008.
2. AREDS: Results National Eye Institute. http://www.nei.nih.gov/amd/background.asp. Accessed February 18, 2014.
3. Evans JR, Lawrenson JG. Cochrane Database Syst Rev. 2012 Nov 14;11:CD000254.
4. Hall H. Antioxidant Supplements for Macular Degeneration. Science-Based Medicine. http://www.sciencebasedmedicine.org/antioxidant-supplements-for-macular-degeneration. Accessed February 18, 2014.
5. Beatty S, Murray IJ, Henson DB, et al. Macular Pigment and Risk for Age-Related Macular Degeneration in Subjects from a Northern European Population. Invest Ophthalmol Vis Sci. 2001 Feb;42(2):439-46.
6. Richer S, Stiles W, Statkute L. Double-masked, placebo-controlled, randomized trial of lutein and antioxidants supplementation in the intervention of atrophic AMD: Veterans LAST study. Optometry. 2004 Apr;75(4):216-30.
7. Richer S, Stiles W, Graham-Hoffman K, et al. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration: The Zeaxanthin and Visual Function Study (ZVF) FDA IND #78, 973. Optometry. 2011 Nov;82(11):667-680.e6
8. Bernstein PS, Zhao D, Wintch SW, et al. Resonance Raman Measurement of Macular Carotenoids in Normol Subjects and in Age-Related macular Degerneration Subjects. Ophthalmology. 2002 Oct;109(10):1780-7.
9. Seddon JM, Ajani UA, Sperduto RD, et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA. 1994 Nov 9;272(18):1413-20.
10. Age-Related Eye Disease Study Research Group; SanGiovanni JP, Chew EY, et al. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch Ophthalmol. 2007 Sep;125(9):1225-32.
11. Wang J, Foran S, Smith W, Mitchell P. Risk of age-related macular degeneration in eyes with macular drusen or hyperpigmentation: The Blue Mountains Eye Study Arch Ophthalmol. 2003 May;121(5):658-63.
12.Blue Mountain Eye Study. Centre for Vision Research. Available at: www.cvr.org.au/bmes.htm. Accessed February 18, 2014.
13. SanGiovanni JP, Chew EY, Clemons TE, et al. The relationship of dietary lipid intake and age-related macular degeneration in a case-control study: AREDS Report No. 20. Age-Related Eye Disease Study Research Group. Arch Ophthalmol. 2007 May;125(5):671-9.
14. Chapman MJ, Ginsberg H, Amaraenco P, et al., Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management Eur Heart J (2011) 32 (11): 1345-1361.
15. Appel LJ, Miller ER 3rd, Seidler AJ, et al. Does supplementation of diet with ‘fish oil’ reduce blood pressure? A meta-analysis of controlled clinical trials. Arch Intern Med. 1993 Jun 28;153(12):1429-38.
16. Hu FB, Bronner L, Willett WC, et al. Fish and Omega-3 Fatty Acid Intake and Risk of Coronary Heart Disease in Women. JAMA. 2002 Apr 10;287(14):1815-21.
17. Simopoulos A. Omega-3 Fatty Acids in Inflammation and Autoimmune Diseases, , J Am Coll Nutr. 2002 Dec;21(6):495-505.
18. Azrad M, Turgeon, Demark-Wahnefried W. Current Evidence Linking Polyunsaturated Fatty Acids with Cancer Risk and Progression. Front Oncol. 2013; 3: 224.
19. Chew EY, Clemons TE, SanGiovanni JP, et al. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013 May 15;309(19):2005-15.
20. Bernstein P, Ahmed F, Aihua L,et al Macular Pigment Imaging in AREDS2 Participants: An Ancillary Study of AREDS2 Subjects Enrolled at the Moran Eye Center. Invest Ophthalmol Vis Sci. 2012 Sep 14;53(10):6178-86.
21. Wang Y, Roger I, Connor SL, et al. Competitive inhibition of carotenoid transport and tissue concentrations by high dose supplements of lutein, zeaxanthin and beta-carotene. Eur J Nutr. 2010 Sep;49(6):327-36.
22. Junod S. FDA + Clinical trials: A short History. FDA. www.FDA.gov./aboutfda/whatwedo/history/overviews/ucm304485.htm. Accessed February 20, 2014.
23. Ma Q, Lu AY. Pharmacogenetics, pharmacogenomics, and individualized medicine. Pharmacol Rev. 2011 Jun;63(2):437-59.
24. Yu Y, Reynolds R, Rosner B, et al. Prospective assessment of genetic effects on progression to different stages of age-related macular degeneration using multistate Markov models. Invest Ophthalmol Vis Sci. 2012;53:1548-56.
25. Seddon JM, Reynolds R, Yu Y, Rosner B. Validation of a prediction algorithm for progression to advanced macular degeneration subtypes. JAMA Ophthalmol. 2013 Apr;131(4):448-55.
26. Awh C, Lane A, Hawken S, et al. CFH and ARMS2 Genetic Polymorphisms Predict Response to Antioxidants and Zinc in Patients with Age-related Macular Degeneration. Presented at: 2013 American Academy of Ophthalmology Annual Meeting; Nov. 16-19; New Orleans, LA.
27. Klein ML, Francis PJ, Rosner B, et al. CFH and LOC3087715/ARMS2 Genotypes and Treatment with Antioxidants and Zinc for ARMD. Ophthalmology. 2008 Jun;115(6):1019-25.
28. Seddon JM, Reynolds R, Maller J, et al. Prediction Model for prevalence and Incidence of Advanced Age- Related Macular Degeneration based on genetic, Demographic and environmental Variables. Invest Ophthalmol Vis Sci. 2009 May;50(5):2044-53.
29. Chakrathy U, McKay GJ, de Jong PT, et al. Arms2 Increases the risk of early and late Age-Related Macular Degeneration in the European Eye Study. Ophthalmology. 2013 Feb;120(2):342-8.
30. Perkins SJ, Nan R, Li K, et al. Complement Factor H- Ligand Interactions: Self Association, multivalency and dissociation constants. Immunobiology. 2012 Feb;217(2):281-97.

