Model Shows Promise for Macular Disease
Human stem cells may open the door for better research—and treatment targets.
By
Without an effective, patient-derived model to work with, researchers have struggled to study macular dystrophies in the laboratory setting. But that might change with a new study in which researchers re-programmed skin cells from patients with three forms of macular dystrophy into stem cells, from which they created retinal pigment epithelium (RPE) cells.1 When aged in a dish, these cells mimicked several characteristics of the disease—effectively giving the researchers a working human stem cell model.1
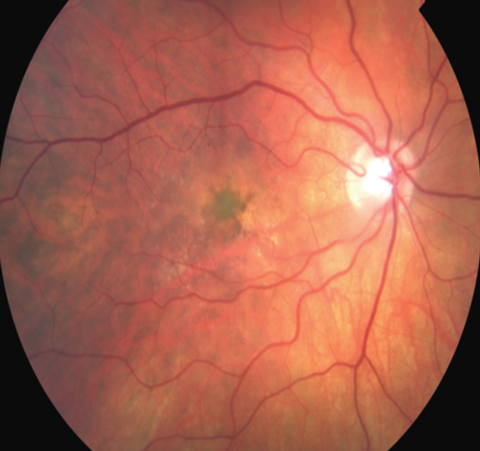 |
| The new model may lead to treatment discoveries for macular dystrophies, such as vitelliform maculopathy. Photo: Virginia Monteith Hodges, OD |
“Now we can actually identify and test a rational drug therapy in patients’ own cells,” Ruchira Singh, PhD, lead author of the study, said in a press release. “So far, this has not been possible, but now we can actually study macular diseases in parallel and identify what might be the central defect across macular diseases.”2
“This study reveals some interesting observations about the role of the RPE in specific similar maculopathies,” says Sara Weidmayer, OD, a clinical assistant professor in the Department of Ophthalmology and Visual Sciences at the University of Michigan.
Already, the model has helped researchers discover that: (1) dysfunctional RPE cells can cause specific aspects of degeneration without the help of other cells or the retina, and (2) a group of molecules within the RPE cells could be targeted by new pharmaceuticals.2
“These discoveries are exciting and may open the door for continued research to further the understanding of the pathophysiologic mechanisms of macular dystrophies and degeneration,” Dr. Weidmayer
says. “Better understanding each retinal cell layer’s role in the development of disease should spur advances that can directly improve patient outcomes in the future.”
1. Galloway CA, Dalvi S, Hung SSC, et al. Drusen in patient-derived hiPSC-RPE models of macular dystrophies. Proceedings of the National Academy of Sciences. 2017:201710430. |
Study: Retina Sheds Light on Alzheimer’s
A new imaging modality in development may help clinicians detect early signs of Alzheimer’s disease (AD). It is well established that the accumulation of amyloid β-protein (Aβ) deposits in the brain may be a biomarker for AD. Currently, brain Aβ is measured with PET scans and cerebrospinal fluid analysis.1
The new imaging technique, relying on fundus autofluorescence (FAF) photography, suggests corresponding deposits of Aβ can be found in the retina. The researchers studied donated eyes and brains of Alzheimer’s patients and controls, as well as high-resolution FAF imaging of Aβ in the eyes of AD patients—the first time technology shows potential to scan living patients, according to the researchers.1,2
They found a 4.7-fold increase in retinal plaque burden in 16 patients with AD, compared with controls, and discovered unique geometric distribution and layer location of amyloid pathology in the retina.1,2 Researchers observed deposits of Aβ in the peripheral regions of the superior quadrant and innermost retinal layers, findings characterized as ‘hot spots’.2 In the future, such high-resolution retinal imaging could be used to identify those at risk for the disease and monitor those who already have a confirmed diagnosis.
Clinical Significance
“On the whole, I am quite encouraged,” says Peter Snyder, PhD, professor of neurology and surgery at Brown University’s Alpert Medical School. It’s still an evolving field, and little consensus exists on the best acquisition techniques and metrics, he says. “But, even if this specific technique is not exactly what we want, in terms of a widely available and easy-to-use screening approach, this shows us that we are on the right track biologically.”
Not everyone agrees with an Aβ-centric approach. “This publication is an example of analytics—using the retina as a ‘potential surrogate marker’ of AD,” says Stuart Richer, OD, PhD, president of the Ocular Wellness and Nutrition Society. “However, tau protein accumulation is likely more important than amyloid, as amyloid is not sufficient itself to bring about AD.” In some cases, centenarians have abundant Aβ on autopsy without documented loss of cognitive function, he says.
The real power of such technologies, he says, is in the future of predictive analytics. “This will involve combining structural data, genetics and environmental information encompassing lifestyle and nutritional factors.” He adds that ODs, who examine 120 million people per year, are key stakeholders in this research. “Optometrists will surely be involved in this emerging worthwhile quest, hopefully by providing counseling, with the goal of educating patients concerning lifestyle choices and behaviors to protect both their eyes and brain.”
1. Businesswire. Clinical study shows that retinal imaging may detect signs of Alzheimer’s disease. www.businesswire.com/news/home/20170822005468/en/Clinical-Study-Shows-Retinal-Imaging-Detect-Signs. Accessed September 7, 2017. |
Apps Detect Disease, Injury Through the Eyes
Researchers are turning to the eyes to better screen for a number of systemic conditions such as concussion and pancreatic, gall bladder and liver dysfunction—with apps.
“Eye doctors are gatekeepers and look to the eye to tell us more about the body,” says Carlo Pelino, OD, assistant professor at PCO at Salus University. While ODs usually catch systemic concerns with dilation, these apps capitalize on ocular effects to the anterior segment.
 |
| The apps currently use a box to control light exposure, but the goal is to launch them without the need for accessories. |
Cancer Screening
An app called BiliScreen uses a photo to detect increased levels of bilirubin in the sclera. When used with a 3D-printed box that controls light exposure, the app successfully identified cases of concern 89.7% of the time, according to the preliminary study.1 Its developers hope the app may one day help improve screening methods for patients at risk for systemic diseases associated with increased bilirubin levels, such as pancreatic cancer.
“Sometimes it can be difficult to detect pancreatic, gall bladder and liver problems until the jaundice of the skin and sclera becomes prominent, which is often too late,” says Dr. Pelino. “But if we can pick that up earlier, that’s huge. That’s an extremely useful tool.”
Currently, bilirubin levels are monitored with a blood test, which can be invasive and inconvenient. The researchers hope the app’s algorithms can detect changes before they are visible to the naked eye, possibly leading to earlier diagnoses and initiation of crucial treatment.
The next step is to test the app on a much larger scale and iron out usability issues such as removing the need for accessories, according to the researchers.
Identifying Concussion
An app known as PupilScreen may help sports professionals quickly identify concussion symptoms on the sidelines.2 The new app is designed to detect changes in pupillary response to light using a smartphone’s video camera and deep learning tools.2 Because traumatic brain injury (TBI) can negatively affect pupillary response, the researchers hope the app will function as an objective screening tool for concussion, mainly for medical personnel on the sidelines.2,3
Also using a 3D-printed box, the app uses the smartphone’s camera and flash to stimulate the patient’s eyes and record a three-second video. The algorithm assesses a patient’s pupillary light reflex, according to the researchers.2 During a pilot study of 48 patients, clinicians identified all six patients within the group who had suffered a TBI.2
“We need something like this to detect concussion quickly, so coaches know a player can’t go back onto the field,” says Dr. Pelino. “Several tools exist, such as the King-Devick test, but this is another piece of the puzzle. We need as many tools as possible on the field to help protect players from the long-term effects of continued injury.”
Dr. Pelino notes the app must be accepted by the neurology community. In addition, he hopes medical personnel are properly trained to use the app and refer to neurology and an eye care professional after positive results.
1. Mariakakis A, Banks MA, Phillipi L, et al. BiliScreen: Smartphone-based scleral jaundice monitoring for liver and pancreatic disorders. PACM Interact Mob Wearable Ubiquitous Technol. 2017;1(2):Article 20. |
FDA to Rein in Stem Cell Clinics
In the wake of several reported incidents of stem cell treatments gone wrong, Food and Drug Administration (FDA) Commissioner Scott Gottlieb, MD, announced August 28, 2017, that the agency is increasing oversight of companies involved in regenerative medicine—an evolving industry often under no obligation to adhere to pharmaceutical regulations.1 While the FDA regulates medical products, many emerging therapies hover between what is regulated and a therapy individualized by a surgeon in such a way that it is no longer subject to FDA regulation.1
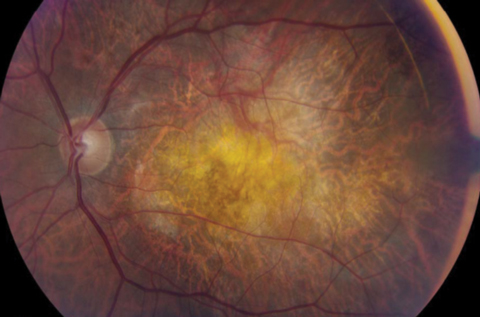 |
| The FDA’s recent stance on stem cell treatment practices is aimed at protecting AMD patients seeking novel treatments. Photo: Mark T. Dunbar |
“There are a small number of unscrupulous actors who have seized on the clinical promise of regenerative medicine, while exploiting the uncertainty, in order to make deceptive, and sometimes corrupt, assurances to patients based on unproven and, in some cases, dangerously dubious products,” Dr. Gottlieb said in a press release.1
To combat this problem, the agency plans to revamp the regulations and ensure they are properly defined, as well as better enforce them to stop clinics from abusing cell-based regenerative medicine and risking patients’ health.
After the agency investigated stem cell clinics in Florida and California, Dr. Gottleib “directed the FDA to launch a new working group to pursue unscrupulous clinics through whatever legally enforceable means are necessary to protect the public health.”1-3
The agency also plans to more clearly define what is and is not regulated and provide manufacturers with a better framework for understanding if approval is necessary for each new product.
Lastly, the FDA will provide a less burdensome path to approval for responsible product developers, which includes individual providers using their own products for regenerative medicine procedures.1
“We can’t let a small number of unscrupulous actors poison the well for the good science that holds the promise of changing the contours of human illness and altering the trajectory of medicine and science,” Dr. Gottlieb emphasized.1
The FDA plans to release documents specifically outlining which new regenerative medical treatments and products are subject to FDA regulation, as well as details on an expedited path for legitimate companies applying for market approval, according to Dr. Gottlieb.2
1. US Food and Drug Administration. Statement from FDA Commissioner Scott Gottlieb, MD, on the FDA’s new policy steps and enforcement efforts to ensure proper oversight of stem cell therapies and regenerative medicine. August 28, 2017. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm573443.htm. Accessed September 6, 2017. |
In the newsThe enzyme leukotriene B4 may be to blame for end-of-day contact lens (CL) discomfort, according to new research. Investigators collected evening tears from 22 symptomatic and 23 asymptomatic CL wearers, then measured the prostaglandins, leukotriene B4 and cysteinyl leukotrienes. They found no significant different in most enzyme levels, with the exception of leukotriene B4, which may play a role in lower comfort scores reported by symptomatic CL wearers. Masoudi S, Stapleton FJ, Willcox MD. Differences in tear film biochemistry of symptomatic and asymptomatic lens wearers. Optom Vis Sci. 2017;94(9):914-8. British researchers found prescribing a supplement that contains antioxidants, zinc and copper may improve the long-term outcomes of patients with wet age-related macular degeneration (AMD). After analyzing data from AREDS, UK treatment costs and AMD prevalence figures for patients older than 55, the researchers calculated that those who take a supplement would need eight fewer injections of anti-VEGF over the course of their lifetime. Lee AY, Butt T, Chew E, et al. Cost-effectiveness of age-related macular degeneration study supplements in the UK: combined trial and real-world outcomes data. British J Ophthalmol. 2017. [Epub ahead of print]. After studying 11 professional and 31 amateur musicians, investigators discovered intraocular pressure (IOP) often rises after playing wind instruments. Although they found similar or even higher IOP levels occurred during other daily activities or at night (as revealed by 24-hour IOP monitoring), they concluded the IOP rise due to playing a wind instrument may play a role in managing glaucoma progression in this subset of patients. de Crom R, Webers C, van Kooten-Noordzij M, et al. Intraocular pressure fluctuations and 24-hour continuous monitoring for glaucoma risk in wind instrument players. J Glaucoma. 2017. [Epub ahead of print]. |

