FDA Approves 24-hour IOP Measuring CL
The device may help clinicians monitor glaucoma progression.
By Bill Kekevian, Senior Editor
In an age of watches that record your heart rate and cell phone apps that track how many calories you burn, it’s only natural that eye care would offer its own contribution to the “wearables” movement. After years of development, the Triggerfish system (Sensimed)—a contact lens designed to continuously measure and record ocular changes closely correlated with intraocular pressure (IOP)—was recently cleared for marketing by the FDA.1
“This device could conceivably be used to determine the pretreatment fluctuations and impact of nighttime IOP on the clinical picture,” says I. Ben Gaddie, OD, president of the Optometric Glaucoma Society. “During treatment, it will help determine true response to medication as well as identify time points when IOP is fluctuating or slipping.”
In addition to the one-time-use silicone contact lens outfitted with a micro-sensor, the kit includes an antenna the patient wears around the eye that wirelessly transfers data from the lens to a portable recording device.
It doesn’t use the traditional millimeters of mercury to evaluate IOP, but Dr. Gaddie says that’s not a problem. He’s interested in using the device to monitor patients who are progressing faster than expected based on in-office IOP measurements. By obtaining a patient’s relative pressure over 24 hours, he says, the device could reveal if “the patient is performing activities that are leading to shorter-term elevation of IOP. Being able to identify these triggers and trends will be of tremendous help to the doctor.”
The Triggerfish will be loaned to patients by a doctor who will use the data to specify treatment, similar to how cardiologists issue temporary heart monitors.
The company seeks to “work closely with the glaucoma community to design and execute a major post-approval study” that will confirm the device’s ability to predict the course of the disease, Senismed CEO David Bailey said in a press release.
|
1. De Moraes C, Jasien J, Simon-Zoula S, et al. Visual field change and 24-hour IOP-related profile with a contact lens sensor in treated glaucoma patients. Ophthalmology. 2016 April;123(4):744-53. |
Cataract Surgery Disparities Revealed
Substantial geographic disparities in cataract treatment are evinced in a new study from the University of Michigan. Recently published in JAMA, the retrospective cross-sectional study used the Clinformatics DataMart database to assess data from 1,050,815 beneficiaries between 2001 and 2011 in 306 US communities.
The study found the median age of patients undergoing cataract surgery for the first time varies by as much as 20 years between some communities—from age 60 in some areas to 80 in others. The variation in the age-standardized cataract surgery rate varies from 7.5% in Honolulu, Hawaii, to 37.3% in Lake Charles, La.
In each of the 306 hospital referral regions included in the study, researchers computed the standard deviations
for the age of cataract surgery recipients, then assessed the timing from an individual’s first recorded cataract diagnosis to the date of the first extraction. The median time between diagnosis and surgery ranges, for example, from 17 days in Victoria, Texas to 367 days in Yakima, Wash.
“With the increasing number of cataract surgeries, optometrists will be called upon to be more involved in the cataract journey from early diagnosis and preoperative evaluation to education and postoperative care,” says Walt Whitley, OD, MBA, director of optometric services at Virginia Eye Consultants. The study “demonstrates the role optometrists have, not just in urban communities, but in rural areas where there are more optometrists available than ophthalmologists.”
In addition to the geographical factor, the researchers took into account age, race, socioeconomic status, level of education, UV light exposure, urban vs. rural residency and the number of ophthalmologists or optometrists in a given community. The data reveals racial disparities persisting, with a reduced likelihood for cataract surgery among black patients.
“The most interesting point is regarding the issues with trust and communication with physicians,” Dr. Whitley says of the study’s look into potential reasons for these racial and socioeconomic disparities. “This is a reminder of the critical role of developing and fostering our relationships with our patients.”
While the study is limited by the information provided in claims data, it makes clear the existence of geographic variation in the timing and rates of cataract surgery among different communities. Furthermore, it highlights the need for further exploration into the underlying causes and the patient impact.
|
Kauh CY, Blachley TS, Lichter PR, et al. Geographic variation in the rate and timing of cataract surgery among US communities. JAMA Ophthalmol. 2016;134(3):267. |
IN THE NEWSAn update to the Diabetic Retinopathy Clinical Research Network Protocol T trial reveals year-two data inconsistent with year-one findings. While year-one data showed Eylea (aflibercept, Regeneron) outperforming both of the other anti-VEGFs for some patients with diabetic macular edema (DME), the year-two results were less conclusive. Researchers found Eylea still outperformed Avastin (bevacizumab, Genentech) at year two, but no longer produced superior results compared with Lucentis (ranibizumab, Genentech). Overall mean VA improvement was 12.8 letters for Eylea, 12.3 for Lucentis and 10.0 for Avastin. New research from Schepens Eye Research Institute found an increase in mitochondrial and nuclear DNA damage in Fuchs’ endothelial corneal dystrophy (FECD) and further correlated this with mitochondrial energy production loss. Identifying the cause of cell death in FECD moves research closer to providing alternative and safer treatments options, Ula Jurkunas, MD, principal investigator, said in a press release. Researchers now hope to focus on developing cytoprotective and anti-aging therapies. We may soon have the technology to regrow lenses and corneas, according to two studies in Nature. The discovery was made using adult cells genetically reprogrammed to an embryonic stem cell–like state. The first study used rabbit models to achieve functional lens regeneration during cataract removal by preserving endogenous lens epithelial stem/progenitor cells. The second study transplanted lab-grown corneas into rabbits born without fully formed corneas. |
Gene Editing May Defeat RP
Scientists used the genomic editing technology CRISPR to correct the gene mutation responsible for retinitis pigmentosa (RP), according to a press release. They used skin samples from patients with RP to generate stem cells that harbored the defective RGPR gene responsible for more than 90% of cases, and then repaired the gene using the bacterial immune system-turned genome-editing tool. By using the patient’s own stem cells, researchers hope they can one day transplant them without a concern for tissue rejection.
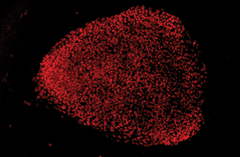 |
| Skin cells were transformed into stem cells, correcting the mutated RPGR gene. Photo: Vinit Mahajan, UI Health Care |
“This particular study proved successful in approximately 13% of the cases,” says Steven Ferrucci, OD, of the US Department of Veterans Affairs in North Hills, Calif., and professor at the Southern California College of Optometry at Marshall B. Ketchum University in Los Angeles. “While this number seems low, it is a great improvement over earlier studies, which had success rates hovering around 1%, demonstrating how far the technology has advanced. Certainly more research needs to be done, but this represents a significant step forward in personalized gene therapy for a wide array of retinal disease.”
| Bassuk AG, Zheng A, Li Y, et al. Precision Medicine: genetic repair of retinitis pigmentosa in patient-derived stem cells. Scientific Reports. 2016 Jan [Epub]. |
Optogenetics Aims to Reverse Blindness
In the first human test of optogenetics—a therapy combining gene therapy and light to control nerve cells—researchers at the Retina Foundation of the Southwest will be injecting DNA from light-sensitive algae into the eyes of patients deemed legally blind from retinitis pigmentosa (RP). The study aims to re-engineer the ganglion cells to respond to light and send signals to the brain, allowing patients to see, if only in blurry black-and-white.
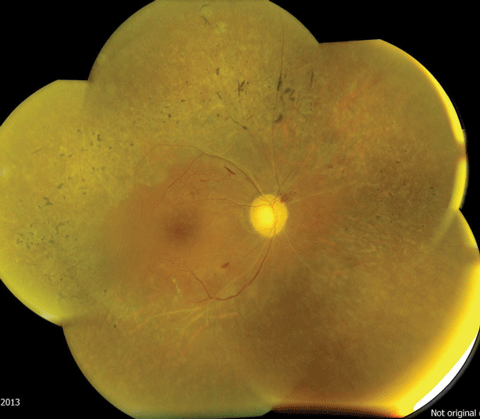 | |
| The pigment spiculing noted along the arcades and the “moth-eaten” retinal degenerative changes are indicative of retinitis pigmentosa. Photos: Mark T. Dunbar, OD | |
| |
The study will enroll as many as 15 patients with retinitis pigmentosa. The researchers hope the new therapy will allow them to see large objects, or even large letters.
But the therapy has its limitations, as the algae protein only responds to blue light. Researchers expect patients to have monochromatic vision, at best. Still, for blind patients to see even this much would be a huge leap forward.
While the new therapy could revolutionize treatment options for patients with RP, it may also have implications for other disease therapies. According to Antonello Bonci, scientific director of the intramural research program at the National Institute on Drug Abuse in Baltimore, researchers will need more information about which cells to target before it can be used for other diseases such as Parkinson’s and severe mental illness. “But that’s five years away, not 20 years away,” he said in a press release.
“We are hopefully getting closer to identifying another treatment option for RP patients who are at the end stage of the disease with bare to no light perception only. Optometrists should share this positivity and hope with their RP patients,” says Ava K. Bittner, OD, PhD, associate professor at Nova Southeastern University. “This clinical trial and a couple of other trials evaluating stem cells for RP are currently recruiting participants in the US, which is exciting since they offer current opportunities for some RP patients who wish to help researchers determine if these approaches might be useful to help improve and/or slow vision loss.”

