We all encounter patients with diabetes in our practice, so it’s incumbent upon us to keep up to date on the extent of its impact and the options available to help, especially in an environment where the research and clinical protocols are rapidly evolving. Retina specialists are busier than ever and expect the referring OD to play a vital role in early identification, patient education and ongoing—possibly lifelong—follow-up. So, let’s make sure we’re on the same page as our syringe-wielding colleagues to ensure patients get the best care possible.
Diabetic eye disease is the leading cause of blindness in American adults, affecting approximately 7.7 million individuals, and the number of people with visual impairment due to diabetic eye disease worldwide is rising—from 2000 to 2010, the number of diabetic retinopathy (DR) cases increased 89%.1,2 Looking forward, the number of patients with DR is expected to double between the years 2010 and 2050.1
Diabetic macular edema (DME) is the leading cause of vision loss and affected approximately 750,000 people in the United States in 2010.3 From 2005 to 2008, an estimated 4.4% of diabetes sufferers had vision-threatening diabetic retinopathy.4 The burden of diabetes-related blindness is not only devastating for the person afflicted, it also costs the United States approximately $500 million annually.5
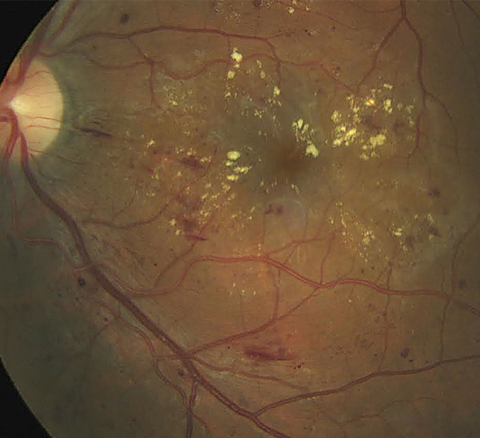 |
| Scattered microaneurysms, intraretinal hemorrhages and nerve fiber layer hemorrhages, along with prominent exudate with clinically significant macular edema. The retinal architecture is partially obscured, indicative of macular thickening. Click image to enlarge. Photo: Erik Hanson, MD |
Drug options
Vascular endothelial growth factor (VEGF) plays a critical role in the pathogenesis of DME and DR. Currently, three primary anti-VEGF molecules are widely used to treat ocular angiogenesis: Avastin (bevacizumab, Genentech), Lucentis (ranibizumab, Genentech) and Eylea (aflibercept, Regeneron). Each debuted in eye care as age-related macular degeneration (AMD) treatments and have since been studied in vein occlusion and DR.
Avastin. This was the first anti-VEGF antibody designed to block all isoforms of VEGF.6 It was approved in 2004 for use in colorectal cancer and subsequently for several other types; it is currently used off-label in the eye.
Lucentis. Genetech believed Avastin would not diffuse through the retina efficiently enough to reach the choroid, so it developed an alternative, shortened molecule, Lucentis.6
Eylea. This recombinant fusion protein functions as a decoy receptor for VEGF-A, developed with improved pharmacokinetics, stronger binding affinity, and a longer half-life than Lucentis and Avastin.6,7
The value of these agents in treating ocular disease continues to be realized and has led to FDA approvals for the treatment of DME and DR in patients with DME and macular edema associated with retinal vein occlusion (Table 1).
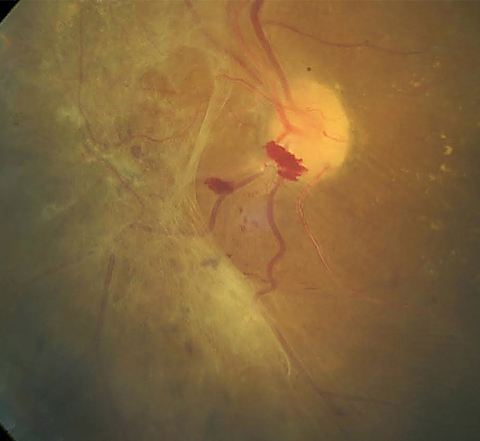 |
| This patient shows a large area of preretinal fibrosis and a small amount of preretinal hemorrhage. The macula demonstrates exudate with macular edema. Click image to enlarge. Photo: Erik Hanson, MD |
The New Gold Rush
Recent years have seen a rapid reappraisal of the treatment protocols for diabetic eye disease, largely due to the success of anti-VEGF options. Long-cherished gold standards of care are being reconsidered, relegated and, in some cases, abandoned.
Diabetic macular edema. Focal laser photocoagulation has long been the standard of care for diabetic macular edema. The Early Treatment Diabetic Retinopathy Study (ETDRS) demonstrates the efficacy of focal macular laser treatment in reducing the risk of moderate vision loss by up to 50% in eyes with clinically significant macular edema.8 However, this classic treatment regimen has now been demoted with the advent of intravitreal anti-VEGF agents. Several recent clinical trials show anti-VEGF therapy (AVT) achieves better visual outcomes vs. focal grid laser for DME.9-14 Though once considered off-label, the FDA recently approved Lucentis and Eylea for DME (Table 1).
While evaluating anti-VEGF agents for their efficacy and safety in DME, the RISE and VISTA studies also noted that diabetic retinopathy has a tendency to improve in patients undergoing AVT.11-14 A second group of researchers demonstrated comparable results, with 39% of eyes improving equal to or greater than two steps on the diabetic retinopathy severity grading system (DRSS) through 36 months in Lucentis-treated eyes.15 In a third instance, similar findings were seen by the Diabetic Retinopathy Clinical Research Network (DRCR), with almost half of the patients in the Lucentis group demonstrating an improvement in DR greater than two steps based on the DRSS.16
Diabetic retinopathy. The gold standard treatment of proliferative DR (PDR) has long been panretinal photocoagulation (PRP), since its evaluation in EDTRS.17 Although there is no denying the efficacy of PRP, it can leave the patient with nyctalopia, reduced contrast sensitivity, visual field constriction and optic atrophy.18 Several studies show Lucentis and Avastin improve PDR without permanent side effects.11-14
In April 2017, Lucentis gained FDA approval for treatment of all forms of DR, including eyes without DME. The approval stems from DRCR’s Protocol S in which Lucentis was compared to PRP for the treatment of PDR. The new Lucentis indication may prove useful for the prevention of visually disabling retinopathy, rather than simply the treatment of it.
Can AVT effectively treat PDR as well as PRP? A recent study sought to investigate this concept. The randomized trial evaluated the superiority of Lucentis vs. PRP for PDR and found no difference in active or regressed neovascularization through two years of follow-up.16
With the FDA approval of Lucentis and Eylea for DR in 2015, the standard of care for this disease is continually evolving. A simple web search on www.clinicaltrials.gov shows an abundance of trials involving AVT, which suggests the progressive nature of research in retinal treatment modalities.19 Many retina specialists are transitioning towards intravitreal injection as the primary treatment modality over laser for a multitude of conditions, according to a worldwide annual survey conducted by the American Society of Retina Specialists (ASRS).20 It found that 86.6% of respondents in the United States would choose either Avastin (62.2%) or Eylea (24.4%) as the treatment of choice in patients with decreased vision due to DME.20 Respondents from all other continents also show a clear preference for AVT in DME, with 60% of respondents choosing AVT as their first-line treatment.20 In addition, the survey found many retina specialists would consider using Lucentis over PRP as the primary treatment for PDR, consistent with results from DRCR’s Protocol S study.16,20 The principal exception: when patient follow-up is unreliable or uncertain.20 Interestingly, only doctors in the United States felt the Lucentis’s cost wasn’t a pertinent factor in determining therapy choice.20
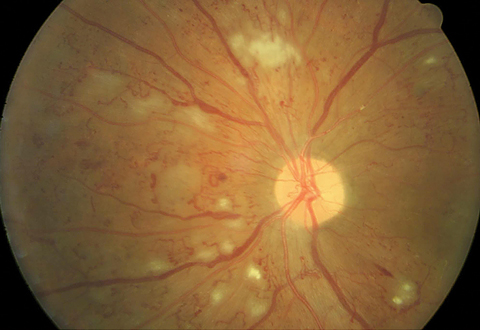 |
| Non-high risk PDR is evidenced by a small amount of neovascularization on the optic nerve. Large amounts of retinal hemorrhages, cotton wool spots and intraretinal microvascular abnormalities (IRMA) are present. Click image to enlarge. Photo: Erik Hanson, MD |
Compare the Costs
A study by the DRCR (Protocol T) comparing Avastin, Lucentis and Eylea for DME showed similar visual acuity outcomes at one year for eyes with baseline acuity of 20/40 or better, but among eyes with baseline VA worse than 20/40, Eylea had a superior (clinically meaningful) visual acuity outcome.22 Through two years of follow up Eyelea remained superior to Avastin, however, there was no longer a significant visual difference when compared with Lucentis.23 Additionally, the two-year results found a minimal difference in the number of injections needed in patients treated with Avastin, Lucentis and Eylea through two years of follow up for DME.22,23
The time has come to talk about the elephant in the room—money. A large price difference exists between the three commonly used AVTs. The 2015 wholesale acquisition costs for single doses of each drug were $60, $1,170 and $1,850 for Avastin, Lucentis and Eylea, respectively.21 As AVTs are often injected multiple times per year, this can result in a significant cost to both the patient and health care system.
Using the above wholesale cost numbers, patient expense in this study would have registered at $960, $17,550 and $27,750 for treatment with Avastin, Lucentis, and Eylea, respectively. Likewise, a post hoc analysis of the DRCR Comparative Effectiveness Trial indicated Eylea and Lucentis are not cost-efficient treatment options relative to Avastin for the treatment of DME.21
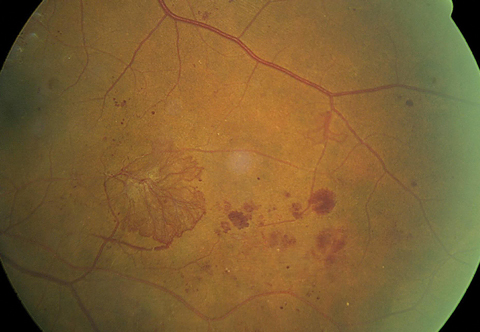 |
| A large area of retinal neovascularization, also known as neovascular elsewhere (NVE), exists. The superotemporal vein demonstrates an omega loop, a strong indicator of retinal hypoxia. Click image to enlarge. |
Consider the Risks
No doubt exists that anti-VEGF therapy is great for treating various posterior segment conditions. However, important drawbacks to the modality exist.
Complications. All of the trials required an intravitreal injection every four to eight weeks (depending on whether Avastin, Lucentis or Eylea was used). Patients treated with AVT for DME or other retinal conditions will often require multiple injections over several months or years. Secondly, AVT is not altogether benign.24 Although serious complications such as endophthalmitis are rare (at approximately 0.028%), they can be visually disabling when they occur.24 Investigators report that ocular side effects of AVTs may include: retinal tears, retinal detachment, vitreous hemorrhage, traumatic iatrogenic cataract, ocular hypertension, ocular hemorrhage and infectious uveitis.24-27
Reported adverse reactions to AVT affect many of the body’s organ systems, including the gastrointestinal, cardiovascular, endocrine, immune and musculoskeletal and excretory systems.23 Of particular interest, a post hoc analysis of the DRCR’s Protocol T study found Lucentis is associated with a higher rate of APTC (Anti-Platelet Trialists Collaboration) events than Eylea and Avastin, including non-fatal strokes and vascular deaths. However, this trend has not been observed in other large randomized control trials including RISE and RIDE.23
While the AVT complication rate is quite low, the vision-threatening complications cannot be dismissed.
Half life. One of the primary detractors from widespread adoption of AVT is the limited half-life. Eylea aimed to change that with improved pharmacokinetics relative to the other two anti-VEGF drugs; however, it still requires dosing every four weeks for the first three months, followed by injections every eight weeks, if needed. Genentech is currently in the process of recruiting patients for a Phase II study that uses a port delivery system (PDS) to deliver Lucentis; the new delivery system aims to allow an ophthalmologist to refill an intraocular device while delivering the drug into the vitreous over an extended time period.28 The study will compare the efficacy of the PDS vs. traditional Lucentis intravitreal injections for patients with AMD-related subfoveal choroidal neovascularization.28 This novel therapeutic approach has the potential to improve the delivery of care not only for AMD, but myriad other retinal conditions, including diabetic eye disease.
We are left with several questions:
• Should diabetic retinopathy be treated earlier with AVT?
• Can we prevent the development of PDR with AVT?
• Is there still a place for laser?
As the face of ocular therapeutics continues to advance, we can expect pharmacologic agents to lead the way in answering these questions and likely change longstanding laser therapy practice patterns.
Table 1. FDA Approval Date and Indication for Lucentis and Eylea30, 31 | ||
| Drug | Date | Approval |
| Lucentis | 2006 | Neovascular macular degeneration |
| 2010 | Macular edema following retinal vascular occlusion | |
| 2012 | Diabetic macular edema | |
| 2015 | Diabetic retinopathy in patients with DME | |
| 2017 | Myopic choroidal neovascularization | |
| 2017 | All forms of diabetic retinopathy | |
| Eylea | 2011 | Neovascular macular degeneration |
| 2014 | Diabetic macular edema | |
| 2014 | Macular edema following retinal vascular occlusion | |
| 2015 | Diabetic retinopathy with macular edema | |
Sit in Your Own Chair
Take a step back and consider your patient’s perspective. You have just been diagnosed with DME and PDR. Would you rather receive a laser treatment that will leave you with permanent side effects or get repeated injections in the eye? Patients should be educated about the available treatment options and the benefits and risks of each. Is the patient reliable for follow up? Does the lack of transportation to appointments pose a concern? Is cost a factor for the patient? These discussions need to begin in the optometrist’s chair. As the delivery methods change and scope of practice advances, we may be deciding the best AVT for our patients.
Dr. Zimbalist practices at Harry S. Truman Memorial Veterans Hospital in Columbia, MO, and is an American Academy of Optometry fellow.
Dr. Scharnweber practices at the St. Cloud VA Health Care System in St. Cloud, MN and is an adjunct faculty at the Pennsylvania College of Optometry.
1. National Eye Institute. Diabetic retinopathy. Available at: nei.nih.gov/eyedata/diabetic. Accessed January 28, 2017. |

