  |
We have all gone to a movie theater and sat through the coming attraction prior to the feature presentation. Some of these clips are quite enticing and leave us anticipating the release of promising entertainment. Similarly, the promise of new therapies fills us with excitement for our profession and patients.
This month, we present a few “coming attractions” in glaucoma pharmacology to pique your professional enthusiasm.
Rhopressa
Rhopressa (Aerie Pharmaceuticals) represents a new class of drug; namely, a Rho-kinase inhibitor. The Rho-kinase family consists of three guanosine triphosphate (GTP)-binding proteins (RhoA, RhoB, RhoC) that regulate aspects of cell shape, motility, proliferation and cellular apoptosis. Research shows inhibition of these kinases relaxes the trabecular meshwork tissues, leading to an enhancement of trabecular aqueous outflow and subsequent IOP reduction.1
Rhopressa, if approved, would be a once-daily product that specifically targets the trabecular meshwork to increase aqueous outflow through this conventional pathway. Currently, only the poorly tolerated miotics exploit this IOP reduction mechanism. Rhopressa is considered “triple action,” as preclinical results demonstrate not only an enhancement of the trabecular outflow, but also a reduction in episcleral venous pressure (which directly contributes to overall IOP levels) and some degree of aqueous production inhibition.2
Despite the seeming potential of this new compound, there have been bumps along the approval road. In a study comparing the compound in Rhopressa (AR-13324 0.02%) with latanoprost, investigators found Rhopressa less effective, by approximately 1mm Hg in patients with elevated IOP.3
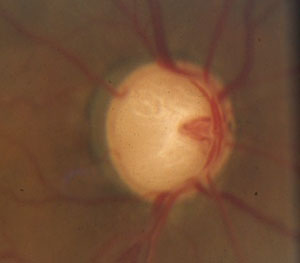 | |
| Advanced glaucomatous damage. New therapies currently being investigated may offer additional IOP-lowering options. |
Additionally, as explained in a press release, Rhopressa did not meet the primary efficacy endpoint of demonstrating non-inferiority to twice-daily dosing of timolol. Rhopressa also showed a slight loss of efficacy over time. However, studies of Rhopressa are ongoing and include a 12-month safety trial with a 90-day interim efficacy assessment and a safety-only study being conducted in Canada.
Roclatan
Another promising agent, Roclatan (Aerie Pharmaceuticals) is a fixed combination agent composed of Rhopressa and latanoprost 0.005%. With the addition of latanoprost to the mix, Roclatan claims to possess “quadruple action,” adding increased uveoscleral outflow to the mechanisms reported by Rhopressa alone. A double-masked, randomized, parallel comparison study compared the IOP-reducing efficacy of the fixed combination Roclatan with its individual components in patients with open-angle glaucoma or ocular hypertension.
The results indicate Roclatan achieved statistical superiority to both latanoprost and Rhopressa, lowering IOP by an additional 1.9mm Hg and 2.6mm Hg, respectively.4
Vesneo
Vesneo (latanoprost bunod, Bausch + Lomb) is a novel nitric oxide-donating prostaglandin F2-alpha analog licensed by Nicox and currently in Phase III clinical development.
In Phase III studies, Vesneo reached its desired primary endpoint of non-inferiority to timolol maleate 0.5% BID, actually showing superiority to the beta-blocker. Vesneo showed a reduction in mean IOP of 7.5mm Hg to 9.1mm Hg from a baseline between two and 12 weeks through Phase III studies.
The VOYAGER study shows latanoprost bunod 0.024% dosed once daily lowered IOP to a significantly greater degree—and had comparable side effects—relative to latanoprost 0.005%. The most common side effect was hyperemia, which was well tolerated.5
Trabodenoson
The compound trabodenoson (Inotek Pharmaceutical) is also in Phase III clinical trials. This compound is a selective adenosine mimetic whose action appears to increase trabecular aqueous outflow.
Trabodenoson stimulates adenosine receptors, increasing metabolic activity in the trabecular meshwork. The increased metabolic activity upregulates proteases that digest and remove accumulated proteins which hinder trabecular aqueous outflow.6 Phase II studies show trabodenoson has a duration of action that may make QD dosing possible. Alone, it approximates the IOP lowering efficacy of prostaglandin analogs.
It also appears to have an additive effect to other, second-line glaucoma medications such as beta-blockers and carbonic anhydrase inhibitors.
Taptiqom
One company may attempt to bring a fixed combination prostaglandin analog/beta-blocker to the United States. The agent, Taptiqom (Santen Pharmaceuticals), combines tafluprost 0.0015% with timolol 0.5% in a preservative-free, unit-dose vial. Early studies are promising. One FDA-approval challenge for fixed combination agents is its inability to demonstrate a significant and sustained efficacy compared with the individual components. A six-month, prospective, randomized, double-masked, parallel group, multicenter Phase III study was performed in patients with ocular hypertension and open-angle glaucoma comparing the fixed combination agent with concomitant use of both components.
The preservative-free tafluprost/timolol fixed combination showed IOP reductions that were both statistically and clinically significant, and non-inferior to those of the concomitant usage of the individual components.
In fact, the fixed combination agent outperformed the concomitant use of both products.7
Similarly, in a study involving exfoliative glaucoma patients, the fixed combination agent performed similarly to the two agents used concomitantly.8
In the past several years, no major developments in topical glaucoma therapy, save the creation of fixed combination agents involving long-used components, have hit the market. These “teasers” promise exciting new innovations in fixed combination agents as well as entirely new medication classes. It remains to be seen if they portend any “blockbusters.”
Drs. Sowka and Kabat have no financial interest in any company or product mentioned.
1. Wang SK, Chang RT. An emerging treatment option for glaucoma: Rho kinase inhibitors. Clin Ophthalmol. 2014;8:883-90.2. Wang RF, Williamson JE, Kopczynski C, Serle JB. Effect of 0.04% AR-13324, a ROCK, and norepinephrine transporter inhibitor, on aqueous humor dynamics in normotensive monkey eyes. J Glaucoma. 2015;24(1):51-4.
3. Bacharach J, Dubiner HB, Levy B, et al. Double-masked, randomized, dose-response study of AR-13324 versus latanoprost in patients with elevated intraocular pressure. Ophthalmology. 2015;122(2):302-7.
4. Lewis RA, Levy B, Ramirez N, et al. PG324-CS201 Study Group. Fixed-dose combination of AR-13324 and latanoprost: a double-masked, 28-day, randomised, controlled study in patients with open-angle glaucoma or ocular hypertension. Br J Ophthalmol. 2015 Jul 24. pii: bjophthalmol-2015-306778.
5. Weinreb RN, Ong T, Scassellati Sforzolini B, et al. VOYAGER study group. A randomised, controlled comparison of latanoprostene bunod and latanoprost 0.005% in the treatment of ocular hypertension and open angle glaucoma: the VOYAGER study. Br J Ophthalmol. 2015;99(6):738-45.
6. Zhong Y, Yang Z, Huang WC, Luo X. Adenosine, adenosine receptors and glaucoma: an updated overview. Biochim Biophys Acta. 2013;1830(4):2882-90.
7. Holló G, Hommer A, Antón López A, Ropo A. Efficacy, safety, and tolerability of preservative-free fixed combination of tafluprost 0.0015%/timolol 0.5% versus concomitant use of the ingredients. J Ocul Pharmacol Ther. 2014;30(6):468-75.
8. Holló G, Ropo A. Intraocular pressure decrease with preservative-free fixed and unfixed combination of tafluprost and timolol in pseudoexfoliative glaucoma. Curr Med Res Opin. 2015;31(1):13-6.

