Amyotrophic lateral sclerosis (ALS)—often referred to as Lou Gehrig’s disease—is a devastating neurodegenerative disease characterized by the loss of cortical, brainstem and spinal motor neurons. The upper and lower motor neurons degenerate or die, disconnecting the communication between the neurons, resulting in progressive muscle weakening and the appearance of fasciculations, or twitching.
Although some patients present with a much slower disease progression, the average period from the onset of symptoms to quadriplegic status is approximately three to five years. We know there should be no autonomic, sensory or cognitive involvement in ALS.1 However, in the later disease stages, up to 50% of ALS patients show cognitive impairment, particularly implicating more severe executive dysfunction and mild memory decline.2
Ocular Involvement
Ocular motor function is largely preserved in ALS. But some evidence shows ocular motor dysfunction in certain individuals diagnosed with ALS in much later stages of the disease.3 The literature is far from rich in ocular symptoms of ALS patients, but ophthalmoplegia has been recorded and was first described in 1925. Although naturally evident in the elderly population, earlier abnormal pursuit has also been described.3 The literature has also shown slowed and inaccurate saccadic movements, especially in the vertical orientation. Individuals with ALS demonstrate an increased error rate and latency to stimuli presented quickly and peripherally. Researchers have also reported horizontal and rotatory gaze-evoked nystagmus. Although less frequently reported, an abnormal Bell’s phenomenon has been seen.3
Vision Demand
Many ALS patients have a special computer-based system that monitors their corneal reflex with an infrared (IR) device. When the patient looks at a letter on their computer keyboard display with both eyes, the IR sensor processes the letter as “typed.” You may see symptoms of computer vision syndrome in these patients. Presbyopia, ocular surface disease and eyestrain, among others, can incorporate many of the symptoms, like headaches, neck/shoulder pain, and blurred vision. More importantly, the IR sensor does not work with a dry ocular surface and is unforgiving, with blinks interrupting a computer action. As a result, ALS patients—with their high visual demands—tend to have partial and reduced blink rate with a resultant ocular dryness.
An Eye on Research
Compared to limb muscles, extraocular muscle (EOM) function is typically well preserved in ALS patients, but is affected in the terminal phase of the disease regardless of type and site of onset. There are two major pathologic findings (through postmortem investigation) in the EOMs of someone with ALS. One finding is an altered cellular architecture of the muscles; the EOMs show hypertrophic and atrophic fibers, increased connective tissue and areas with fatty replacement. The second finding is altered muscle protein content within the EOM fibers.4 In limb muscles of adults, there can be loss of up to 80% of muscle mass and atrophy of individual muscle fibers as a result of denervation.5 The EOMs likely react differently to denervation because they actually show preservation of the muscle fibers.4 The difference in muscle cell communication in EOM and in limb muscle suggests a major role in the pathophysiology of ALS.5
Epidemiology
ALS is one of the most common motor neuron degenerative diseases and typically affects adults during midlife, with a median age onset of 60 years.6 Approximately 5% to 10% of cases are the familial form of the disease (FALS), while the majority of ALS cases are sporadic (SALS) in nature. The onset age of FALS is a decade earlier than the onset age of SALS. The overall incidence of ALS is about three people per 100,000 a year.6
Diagnostic Considerations
Diagnosis of ALS is challenging because many motor neuron diseases share similar symptoms, indicating a functional loss of upper and/or lower motor neurons.7 Electrodiagnostic testing, imaging, laboratory testing and other procedures are necessary to categorize a patient as having a neuromuscular disease. (See “Tests to Diagnose Neuromuscular Disease,” below.) The diagnosis of ALS is largely dependent on differentiation between these diseases and what symptoms are present.
|
Tests to Diagnose Neuromuscular Disease
| |
|
Electrodiagnostic testing |
Electromyography (EMG) |
|
|
Nerve conduction velocity (NCV) |
|
Imaging |
X-rays |
|
|
Magnetic resonance imaging (MRI) |
|
|
Myelogram of cervical spine |
|
Laboratory testing |
Blood serum • High resolution serum protein electrophoresis • Thyroid hormone levels • Parathyroid hormone levels |
|
|
Urine studies • 24-hour heavy metal collection |
|
Spinal tap |
Analysis of cerebral spinal fluid |
Early clinical manifestations of ALS appear gradually over several years. The motor neurons of four different anatomical regions are affected:
• Bulbar—jaw, face, palate, tongue and larynx
• Cervical
—neck, arms, hands and diaphragm
• Thoracic
—back and abdomen
• Lumbosacral
—the lower back, buttocks, legs and feet
ALS is often classified by its site of onset, which typically falls into two categories: bulbar and limb, which includes the cervical, thoracic and lumbosacral regions.
Bulbar implies poor prognosis and shorter life expectancy than limb onset because it affects vital functions like breathing and swallowing. With limb onset, paralysis begins at the limbs and proceeds proximally.8
A Pause for Cause
While the pathogenesis of ALS is unknown, evidence suggests that excessive stimulation of the motor nerve cells by the neurotransmitter glutamate damages and destroys them, which may play an important role in the disease.7
Histopathological studies have revealed foreign deposits within surviving motor neurons, extensive gliosis and the presence of extensive neuronal loss or atrophy.9
Additionally, environmental risk factors and causes of ALS are considered controversial; however, there is significant literature linking the disease to welding and soldering, inhalation of lead vapor, agricultural chemical exposure and electrical trauma. The most common associations are age (between 50 and 80 years), sex (1.5 times greater in males) and physical injury, which may explain the higher incidence in men.7
Treatment
There is no cure for ALS at this time. The best treatment for the patient is to maintain independence through comprehensive and holistic care. The only disease-specific treatment currently available is the neuroprotective agent Rilutek (riluzole, Sanofi-Aventis). In two randomized placebo-controlled double blind trials, Rilutek was shown to slow the rate of muscle strength deterioration and increase the survival rate of the individual.10
Case Report: A New ‘Type’ of Life
We present a patient with a unique chief complaint presentation and attempt to find our way towards successful treatment of his symptoms. This case report outlines various concepts that each eye doctor should consider when examining a patient with ALS.
• History. Our patient is a 55-year-old white male veteran who presented as a low vision patient, with his last dilation performed approximately a year ago. He was diagnosed with ALS more than seven years prior and communicated his complaints through the following printout that he prepared for us:
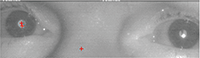
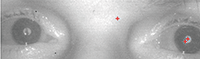
“These are photos of my eyes taken in IR. Tobii’s concern is the shadows on the pupils of the eyes. They are saying that even though the shadows aren’t consistent, they could disrupt the algorithms that track my eyes. Is there any medical cause for the shadows?”
| |
| |
| Our patient types using infrared (IR) eye-tracking software (Tobii). The red crosses in the pupil indicate that the IR system recognizes his eye. Notice the shadow corresponds with no red crosses. |
The patient has quadriplegia. He travels in a motorized wheelchair that his wife operates with a controller. He depends on his wife and home care nurse to suction his saliva (because he cannot swallow), scratch a facial itch or wipe the perspiration on his forehead. A ventilator provides him the ability to breathe.
His only controlled muscles are in his face. In limited observation, it appears he has full EOM range of motion, blinks completely and has a few functional mouth muscles that allow him to occasionally mouth words. He raises his frontal muscles to communicate in a positive manner to a question. All other facial muscles appear paralyzed.
He was an IT guru prior to his health-related tangent. He has his own email and Facebook accounts, and even created his own YouTube channel. It’s quite obvious from his chief complaint that he is cognitively present, despite how his appearance may present.
• Diagnostic data. We considered him a low vision patient based on his complaint, the need for a longer examination time, and the fact that he had a dilated fundus exam within the last year. He does not fit the typical low vision patient protocol. We took his complaint very seriously, because his computer system (Tobii I15, Tobii Technology) is how he communicates. A few times, we observed some fairly dramatic exchanges of miscommunication. This proved the importance of being able to use his computer system optimally for communication.
Retinoscopy to assess the media quality showed a very bright and strong reflex both eyes. Visual acuity was approximately 20/25 with a slightly hyperopic subjective refraction of +0.50D OD and +0.25 D OS at distance. At near, we performed a trial lens refraction and found that +1.75D add would allow best vision for his computer, which was attached to an arm of his motorized wheelchair. He was extremely pleased with his vision potential.
When we discussed that he would need a computer prescription to use his Tobii, it was relayed to us that he also looks at the television to his right, which is located about 15 to 20 feet away. Because he was not able to move his head at all, this created an interesting dilemma as a single vision lens would not allow him to do his two most frequent activities of daily living (ADLs).
We measured his pupillary distances and helped him pick out a frame. We called an optical lab and worked out that a “lined side-focal” with 45mm segment centered within the frame would be his best bet. A few days later, the optical lab contacted us stating they could not fill the order since the lens blank size would not allow for the measurement specifics. The lab sent us glasses with the best they could do. Additionally, we requested that they send us distance vision-only glasses, which allowed us the option of “stick-on” bifocal segments as another alternative. The lab did mention that they could glue two prescriptions together, similar to an executive lens, but we didn’t pursue this option.

|
|
|
Patient’s view through the “lined side-focal.” Note that it was designed for computer distance with central clarity, while also allowing sharp vision to the right for watching TV.
|
• Diagnosis. Our patient was diagnosed with computer vision syndrome (CVS) encompassing dry eye syndrome and presbyopia.
• Treatment and follow-up.
We prescribed preservative-free artificial tears every hour and educated him about the need for more frequent, full blinks.
A month later, he came in to receive and evaluate his two pairs of glasses. His wife reported improvement in his eyes not being nearly as red. She also noticed that at times he blinks more completely. We performed an Ocular Surface Disease Index (OSDI) survey, which resulted in a diagnosis of “mild dryness” with a score of 20.45 (0.0 = no dryness to 100.0 = severe dryness). His most significant complaint was grittiness. It’s worth noting that, at this visit, he was given a relaxation medication (lorazepam) that allowed him to travel better. Unfortunately, it had side effects of drying his eyes out, making his fissures reduced and causing fatigue.
Eye Tracking Software
The patient’s Tobii eye-tracking device uses image-processing algorithms and mathematical models to map the human eye. The feature that primarily pertains to our patient’s complaint is what Tobii Technology refers to as the “eye-tracking optics technology,” which uses optics systems with proprietary production and calibration equipment that is optimized specifically for eye tracking.
Contacting the company provided only limited information as they were understandably protective of their technology. Our best hypothesis on how the eye tracking works is off the corneal reflex and the center of the pupil, which would answer the patient’s question about the shadows and our discovery of his ocular surface dryness.
In the patient’s chief complaint picture, the red “+” signs mark the center of the pupil and the corneal reflex. We further postulate that both of these need to be present and detected in each eye before the system will register the command. Both of these points direct us toward ocular surface disease. If the corneal reflex is muted, it alludes to an unstable tear layer. If the tear layer is unstable, it will also affect the clarity of the IR system to see where the center of the pupil would be. Our patient was more concerned about the computer picking up his eye movement rather than any ocular complaints.
Computer Vision Syndrome
Computer vision syndrome (CVS) describes a group of eye and vision-related problems that result from prolonged computer use. Symptoms vary from eyestrain, headaches and neck/shoulder pain to blurred vision and dry eyes. Such symptoms may be caused by any single cause or combination of the following: poor lighting; glare on the screen; improper viewing distances; poor seating posture; uncorrected refractive error; reduced and incomplete blink rate; and poor tear layer stability secondary to surrounding environmental conditions.11
Vision problems of uncorrected hyperopia, astigmatism and presbyopia can be major contributors to computer vision syndrome. Often, visual symptoms are temporary and improve after stopping computer work. Prevention or reduction of vision problems associated with CVS involves taking steps to better control lighting and glare off the screen, proper working distance and posture for computer viewing, avoiding direct air flow and increasing room humidity, and addressing even minor refractive error, including presbyopic needs. In addition, take steps to treat symptoms as they present, including the use of ocular lubricants and abiding by the 20/20/20 rule—taking at least 20 seconds to look at least 20 feet in the distance after every 20 minutes of computer use.
Although our patient was not symptomatic for pain or strain, ergonomic factors likely play a role in the functioning of Tobii, the eye tracking software, and consequently affect his ability to communicate and function in his activities of daily living. He definitely had symptoms of blurred vision and dryness, which pertained to his chief complaint. The inability of the IR tracking program to find a corneal light reflex points toward an ocular surface problem directly related to extensive computer use.
Take note that our patient only brought up the most significant complaint—the shadows found on his eye-tracking software—but he most likely has additional, less significant complaints systemically. For instance, he had never communicated to us that his eyes often felt gritty until he revealed it in the OSDI survey. This emphasizes the importance of the OSDI survey for our patient and others like him. The survey questions are easy for patients to answer and unveil plenty of information that otherwise may not be discovered.
Working Distance
Due to his extensive IT work history, it is not surprising that our patient has multiple options for computers. He has a travel Tobii that mounts on an arm attached to his motorized wheelchair. When he is home, he has a different desk system that is more extensive. We measured his near reading prescription for the travel Tobii and we were informed that the home system could have the same distance. The patient and his wife understood the need for consistent distance with any type of reading prescription he would use.
To assist the patient in his need for both distance and near correction, we had to solve the problem of providing clear vision at a near distance for the communication board, as well his distant television. Normally, this would be achieved through a bifocal or progressive style lens. Unfortunately, the standard bifocal segment does not center on the patient’s communication board, where the vast majority of his ADLs occur. A pair of “near only” glasses would solve this problem, but offer no help for distance viewing of the television. The progressive lens would have the same problem, and also potentially complicate how the IR tracking software would sense eye movements behind this style of lens. Therefore, our solution had to overcome the standard positioning of an add power while maintaining an area (specifically the upper right) of the lens periphery for distance viewing.
Our first attempt was to have a lab specially cut and decenter a lens to move the add to the center of the frame. This idea could not be accomplished, though, because there was not enough lens blank to compensate for such a shift in the add positioning, the lab explained.
Our second attempt was to use a flat-top bifocal—a flat-top 45 (FT45)—and turn it sideways (we termed this a “lined side-focal”). Our goal was to center the lined side-focal within the lens to allow for clear distance vision in the periphery of the lens. This proved less than ideal because, again, the lab was limited by the lens blank and couldn’t center the FT45 in the frame according to the pupillary distance requested. This change in pupillary distance resulted in an induced prism, which caused diplopia. That said, this attempt showed some promise that such an add could be beneficial.
The third attempt was exactly the same as the second, but using stick-on add lenses over a distance-only pair of glasses. This better centered the add in the frame and the patient seemed pleased with his vision and lack of diplopia. However, the IR tracking software for the communication board did not pick up the patient’s eye movements well. So, for communication purposes, the patient felt that this was not the better of the two side-focal options.
Increase Blink Rate
We also observed during the clinical visits that our patient had a decreased blink rate. In a study analyzing blink rate patterns, subjects were videotaped in a standard setting while performing three different tasks: resting quietly; reading a short passage; and talking freely.12
The average blink rate was then calculated from the data. At rest, the average blink rate was 17 blinks/minute. During conversation, it increased to 26, but it was as low as 4.5 while reading.
We grossly observed our patient to have a blink rate between 1 to 1.5 blinks/minute. Involuntary blinking is, in large part, controlled by the caudate nucleus in the brain and is thought to be influenced by the amount of dopamine present.12 Another large component is the ocular surface condition of the eye and the sensory signals sent when dryness or irritation is felt. Also working against our patient is that he worked in IT, spending long hours on computers for many years, which may have served to desensitize his corneal nerve endings. The patient has also unintentionally, but to some degree necessarily, trained himself to blink less because the cursor on his communication board is lost every time he blinks his eyes.
The reduced blink rate likely relates to the biggest issue we noted from his OSDI survey: a significant complaint of grittiness. Although the OSDI survey picked this up, our patient never it saw as a big enough issue to bring to our attention. This showed us that he most likely lives with many complaints, but is very selective about which ones he actually reports during his many medical health exams.
However, the reduced blink rate does directly relate to the variable shadows detected on his computer system. The shadows seen on the printout complaint were from his tears breaking up. Therefore, we started the patient on artificial tears and, afterward, the patient’s wife reported that his redness was much better. We also wanted to encourage good blinking habits consisting of long, full blinks when the patient remembers, and especially before the IR tracking action is initiated during calibration of the communication board.
Additional Disruption
In addition to his ocular surface condition interfering with his ability to use his communication board, he has other, lesser known factors/complications consistent with individuals afflicted with ALS.
A common late-stage symptom—linked to the degeneration of facial neurons and evident during his clinic visits—is pruritus of the nose and face. The patient’s wife and attending nurse always have a towel at hand to wipe/scratch the nose and face of our patient. The towel serves a dual purpose, as another late-stage symptom is excessive oil secretion from facial pores. Several times while we worked with him he had requested the towel to wipe excess oil from his face. The symptoms are aggravating to the patient and wiping of the face is disruptive to the eye-tracking software, which consequently disrupts communication.
Other factors to consider are saliva suction of the mouth and tracheotomy tube to avoid aspiration, and adding and removing blankets to control body temperature. It is difficult to say if all of these symptoms are disease related or pharmaceutically induced from treating other symptoms; nonetheless, they play a significant role in the disruption of his communication.
Troubleshooting to 20/Happy
Along the journey of finding a pair of glasses that would allow distance vision (to see his TV on the right) and near vision (to use his Tobii central and left), we found that certain options did not work. When our patient returned to assess his abilities with the new glasses options, he was medicated with an anti-anxiety drug (lorazepam) for travel. Unfortunately, this medication also caused our patient to tire easily. We had to rely on him to test out the various options at home. He found that neither pair of glasses worked well, with the stick-on bifocals being the least beneficial. The FT45 lined side-focal seemed to be better but, because of the restrictions on where the lens could be placed, it caused some diplopia when he looks to the left. We will order a specialty pair of glasses with more exact measurements if we feel it would allow him to succeed.
Our patient randomly tried over-the-counter polarized sunglasses and a separate pair of high-end polarized sunglasses with anti-reflective coating. To our surprise, he reported that the OTC option was more effective. Still, with sunglasses on, the patient would have to increase the brightness of the computer monitor in order to see the screen as well.
Variables that may affect the outcome of our patient’s use of his Tobii communication unit include:
• Glasses parallel to the computer screen. The eyeglasses must be as parallel to the computer screen as possible. The photo above shows the chief complaint. You can see that the two red “+’s” are located on the corneal reflex and the center of the pupil. Would the IR sensors read the eye movements better at a certain viewing angle?
• Anti-reflective coating
. ARC reduces light reflections and allows the patient to see better out of the lenses. Would ARC dim or mute the corneal reflex for the IR sensor to read?
• Polarized lenses
. Does the quality of polarization help or hurt? Does polarization allow the patient to see better through the lenses and the IR sensor to see the patient’s eye better through the lens, too? Could too much polarization, or higher quality polarization, prevent the IR sensor from getting accurate results?
• Lens material
. Would lower Abbe values prevent the IR sensor from being as accurate?
• Other add-on coatings
. Would any other type of coating or add-on, such as progressive addition lenses, affect the reading ability of the IR sensor?
• Pupil size.
With sunglasses, the light is shaded and the pupil gets bigger. We are unsure how the pupil size would have any effect on the IR sensors. The consultant from Tobii Technology mentioned that pupil size may be a variable that needed to be controlled. When questioned, he had no reasoning behind that thought.
• Corneal reflex. A dry cornea mutes the reflex.
Airflow and Room Humidity
Airflow within the patient’s room also had to be addressed because direct airflow within the house increases the rate of evaporation of tears from the ocular surface. His room has a ceiling fan that is always running, so we suggested using an indirect floor fan to provide airflow in its place. We also recommended a room humidifier to increase air moisture within the house.
Conclusion
We worked diligently on addressing the patient’s chief complaint, which was essentially ocular surface disease.
First, we had to educate the patient, his wife and his home care aide. We provided information regarding how ocular surface dryness could cause not only ocular discomfort and redness, but poor vision and inability for the eye-tracking software to properly pick up the IR reading-signal from of communication board. A proper spectacle correction would allow for sharper vision, likely reducing strain and allowing for viewing of multiple distances. We also had to emphasize healthy blinking habits. The importance of regular, full blinks was not routine him, and may not have been for some time given his previous employment in IT. Our goal was for him to achieve a healthier and clearer refractive surface, allowing for clearer vision, better accuracy and more ocular comfort while maintaining his ability to communicate and function on a daily basis. In addition, we instructed his wife and health aide to use artificial tears on a regular basis to keep his eyes moisturized. (Ointment was not a consideration because it would cause blurring and disrupt his ability to communicate.)
This unique case report describes a patient with ALS who presented with complaints of his computer software detecting issues with his eyes, which prevented him from his only way to communicate. After assessing his visual demands, expectations and ADLs, we considered many options and tried to provide successful treatment. Further, ocular lubrication, reduction of direct airflow and increased blink rate allowed his ocular surface become more stable, which directly improved his communication ability.
We achieved various improvements for our patient, and learned a few lessons ourselves. He is doing better now than when we first met him. We will figure out the right mix of optical correction for him to maintain maximum vision at his two main distances to perform his ADLs.
Here is what the patient wrote after his treatment: “Many ALS patients rely on their eyes for communication and [my wife] and I have always felt that as long as I can communicate, I will stick around. Your work is literally saving my life and allowing me to have a reasonable quality of life. Thank you for that.”
Dr. Olsen is the current resident at the Ralph H. Johnson VA Medical Center in Charleston, SC. Dr. Denton is his residency director.
1. Brooks BR, Miller RG, Swash M, Munsat TL, World Federation of Neurology Research Group on Motor Neuron Diseases. El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord. 2000 Dec;1(5):293-9.
2. Rusina R, Ridzon P, Kulist'ák P, et al. Relationship between ALS and the degree of cognitive impairment, markers of neurodegeneration and predictors for poor outcome. A prospective study. Eur J Neurol. 2010 Jan;17(1):23-30.
3. Sharma R, Hicks S, Berna CM, et al. Oculomotor dysfunction in amyotrophic lateral sclerosis: a comprehensive review. Arch Neurol. 2011 Jul;68(7):857-61.
4. Ahmadi M, Liu J-X, Brännström T, et al. Human extraocular muscles in ALS. Invest Ophthalmol Vis Sci. 2010 Jul;51(7):3494-501.
5. McLoon LK, Harandi VM, Brännström T, et al. Wnt and extraocular muscle sparing in amyotrophic lateral sclerosis. Invest Ophthalmol Vis Sci. 2014;55(9):5482-96.
6. Wijesekera LC, Leigh PN. Amyotrophic lateral sclerosis. Orphanet J Rare Dis. 2009;4:3.6.
7. Charles T, Swash M. Amyotrophic lateral sclerosis: current understanding. J Neurosci Nurs. 2001 Oct;33(5):245-53.
8. Louwerse ES, Visser CE, Bossuyt PM, Weverling GJ. Amyotrophic lateral sclerosis: mortality risk during the course of the disease and prognostic factors. The Netherlands ALS Consortium. J Neurol Sci. 1997 Oct;152 Suppl 1:S10-7.
9. Calvo AC, Manzano R, Mendonça DMF, et al. Amyotrophic lateral sclerosis: A focus on disease progression. Biomed Res Int. 2014;1-12.
10. Lacomblez L, Bensimon G, Leigh PN, et al. A confirmatory dose-ranging study of riluzole in ALS. ALS/Riluzole Study Group-II. Neurology. 1996 Dec;47(6 Suppl 4):S242-50.
11. Zheng Y, Liang H, Hao C, Lu F. Computer vision syndrome: a widely spreading but largely unknown epidemic among computer users. Computers in Human Behavior 2008 Sep;24(5):2026–2042.
12. Bentivoglio AR, Bressman SB, Cassetta E, et al. Analysis of blink rate patterns in normal subjects. Mov Disord. 1997 Nov;12(6):1028-34.

